
Filed Pursuant to Rule 424(b)(5)
Registration No. 333- 217106
PROSPECTUS SUPPLEMENT
(To Prospectus dated April 12, 2017)

27,272,727 Shares of Common Stock
Matinas BioPharma Holdings, Inc.
We are offering shares of our common stock.
Our common stock is traded on the NYSE American LLC, or NYSE American, under the symbol “MTNB”. On March 15, 2019, the last reported sales price of our common stock on NSYE American was $1.30 per share.
Investing in our common stock involves risks that are described in the “Risk Factors” section beginning on page S-24 of this prospectus supplement.
We are an “emerging growth company” as defined by the Jumpstart Our Business Startups Act of 2012.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus supplement and accompanying prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
| Per Share | Total | |||||||
| Public offering price | $ | 1.100 | $ | 30,000,000 | ||||
| Underwriting discounts and commissions (1) | $ | 0.066 | $ | 1,800,000 | ||||
| Proceeds to us, before expenses | $ | 1.034 | $ | 28,200,000 | ||||
(1) See “Underwriting” beginning on page S-60 in this prospectus supplement for a description of the compensation to be paid to the underwriters in connection with this offering.
The underwriters may also exercise their option to purchase up to an additional 4,090,909 shares from us at the public offering price, less the underwriting discounts and commissions, for 30 days after the date of this prospectus supplement. If the underwriters exercise the option in full, the total underwriting discounts and commissions payable by us will be $2,070,000 and the total proceeds to us, before expenses, will be $32,430,000.
The underwriters expect to deliver the shares against payment therefor on or about March 19, 2019.
BTIG, LLC
The date of this prospectus supplement is March 15, 2019.
TABLE OF CONTENTS
Prospectus Supplement
| Page | |
| ABOUT THIS PROSPECTUS SUPPLEMENT | S-ii |
| SUMMARY | S-1 |
| THE OFFERING | S-23 |
| RISK FACTORS | S-24 |
| SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS | S-56 |
| USE OF PROCEEDS | S-58 |
| DILUTION | S-59 |
| UNDERWRITING | S-60 |
| LEGAL MATTERS | S-63 |
| EXPERTS | S-63 |
| WHERE YOU CAN FIND MORE INFORMATION | S-63 |
| INCORPORATION OF CERTAIN INFORMATION BY REFERENCE | S-64 |
Prospectus
| S-i |
ABOUT THIS PROSPECTUS SUPPLEMENT
In this prospectus supplement, “Matinas,” “we,” “us,” “our” or “ours” refer to Matinas BioPharma Holdings, Inc. and its consolidated subsidiaries.
This document is in two parts. The first part is this prospectus supplement, which adds to and updates information contained in the accompanying prospectus. The second part, the prospectus, provides more general information, some of which may not apply to this offering. Generally, when we refer to this prospectus, we are referring to both parts of this document combined. To the extent there is a conflict between the information contained in this prospectus supplement and the information contained in the accompanying prospectus, you should rely on the information in this prospectus supplement.
This prospectus supplement and the accompanying prospectus relate to the offering of shares of our common stock. Before buying the shares of common stock offered hereby, we urge you to carefully read this prospectus supplement and the accompanying prospectus, together with the information incorporated herein by reference as described under the headings “Where You Can Find More Information” and “Incorporation of Certain Information by Reference.” These documents contain important information that you should consider when making your investment decision. This prospectus supplement contains information about the common stock offered hereby and may add, update or change information in the accompanying prospectus.
You should rely only on the information that we have provided or incorporated by reference in this prospectus supplement and the accompanying prospectus. Neither we nor the underwriters (or any of our or its respective affiliates) have authorized any other person to provide you with different information. If anyone provides you with different or inconsistent information, you should not rely on it.
We and the underwriters are not making offers to sell or solicitations to buy our securities in any jurisdiction in which an offer or solicitation is not authorized or in which the person making that offer or solicitation is not qualified to do so or to anyone to whom it is unlawful to make an offer or solicitation. You should assume that the information in this prospectus supplement and the accompanying prospectus or any related free writing prospectus is accurate only as of the date on the front of the document and that any information that we have incorporated by reference is accurate only as of the date of the document incorporated by reference, regardless of the time of delivery of this prospectus supplement, the accompanying prospectus or any related free writing prospectus, or any sale of a security.
This prospectus supplement and the accompanying prospectus contain summaries of certain provisions contained in some of the documents described herein, but reference is made to the actual documents for complete information. All of the summaries are qualified in their entirety by the actual documents. Copies of some of the documents referred to herein have been or will be filed as exhibits to the registration statement of which this prospectus is a part or as exhibits to documents incorporated by reference herein, and you may obtain copies of those documents as described below under the headings “Where You Can Find More Information” and “Incorporation of Certain Information by Reference.”
| S-ii |
The following summary of our business highlights some of the information contained elsewhere in or incorporated by reference into this prospectus supplement. Because this is only a summary, however, it does not contain all of the information that may be important to you. You should carefully read this prospectus supplement and the accompanying prospectus, including the documents incorporated by reference, which are described under “Incorporation of Certain Information by Reference” and “Where You Can Find More Information” in this prospectus supplement. You should also carefully consider the matters discussed in the section titled “Risk Factors” in this prospectus supplement, in the accompanying prospectus and in other periodic reports incorporated by reference herein.
Our Company
We are a clinical-stage biopharmaceutical company focused on creating value through (i) the streamlined development under Section 505(b)(2) of the Federal Food, Drug and Cosmetic Act, or FDCA of our lead product candidate, MAT9001, a highly purified, prescription-only omega-3 free fatty acid formulation specifically designed for the treatment of cardiovascular and metabolic conditions and (ii) the application of our lipid nano-crystal (LNC) platform delivery technology to solve complex challenges relating to the delivery of small molecules, gene therapies, vaccines, proteins and peptides. In general, the development timeline for a 505(b)(2) New Drug Application, or NDA, is shorter and less expensive than an NDA developed under Section 505(b)(1) for new chemical entities that have never been approved in the United States. Based upon MAT9001’s unique mixture of highly purified omega-3 free fatty acids and our observations of MAT9001’s enhanced bioavailability and potency as compared to Amarin Corporation’s Vascepa® (icosapent ethyl) in our initial head-to-head pharmacokinetic (PK) and pharmacodynamic (PD), or PK/PD, clinical study, we believe that the results of our forthcoming targeted clinical development activities and related clinical investigations may yield an improved therapeutic profile compared to currently-existing therapies.
MAT9001 is a soft gelatin capsule containing a complex mixture of polyunsaturated free fatty acids, including multiple long-chain omega-3 fatty acids, including primarily eicosapentanoic acid (EPA) and docosapentanoic acid (DPA). Amongst other properties, omega-3 fatty acids, which are also found in FDA-approved drugs in varying amounts, such as Vascepa®, have extensive clinical evidence of safety and efficacy in lowering triglycerides (TG) in patients with hypertriglyceridemia (HTG). We believe that based upon MAT9001’s unique composition, which includes more DPA than other known omega-3 fatty acids, it will prove to be differentiated from other existing therapies for the treatment of very high triglycerides, or severe hypertriglyceridemia (SHTG), and dyslipidemia.
Triglycerides are fats that are carried in the blood, together with cholesterol, within lipoproteins. High levels of triglyceride-rich lipoproteins are associated with an increased risk of atherosclerotic cardiovascular disease and in the case of severe hypertriglyceridemia, acute pancreatitis. High levels of triglycerides are due to both genetic and environmental factors and are associated with comorbid conditions such as diabetes, chronic renal failure, and nephrotic syndrome. Unlike the currently approved products in this category, many of which have been repurposed following clinical failures in their originally intended indications, we have specifically designed and developed MAT9001 to treat SHTG, dyslipidemia and other cardiovascular and metabolic conditions.
In 2015, we announced results from our head-to-head PK/PD clinical study against Vascepa, a prescription-only ethyl ester formulation of EPA. This was an open-label, cross-over design study conducted in 42 patients with elevated triglyceride levels. Patients were treated for 14 days with MAT9001 or Vascepa, (4 grams/day for both treatment arms), followed by a five-week wash-out period, then crossed over to the other treatment. The main objectives of the study were to measure the relative bioavailability of MAT9001 versus Vascepa as well as effects on triglyceride levels. In this study, we observed statistical superiority of MAT90001 in reducing serum triglycerides, total- and non-HDL-cholesterol, apolipoprotein CIII and PCSK9 levels. MAT9001 was observed to significantly reduce PCSK9 in patients. In this trial, MAT9001 achieved greater median percentage reduction in four of six lipid measures, including total cholesterol, when compared to Vascepa.
We are focusing our initial efforts on developing MAT9001 with an initial indication for the treatment of severe hypertriglyceridemia. If we receive U.S. Food and Drug Administration (FDA) approval for severe hypertriglyceridemia, we may seek approval for use of MAT9001 in additional indications, including the treatment of patients with mixed dyslipidemia who are already undergoing treatment with a statin, a commonly used class of cholesterol-lowering medications.
| S-1 |
Consistent with our strategy first put in place when we filed the investigational new drug (IND) application for MAT9001 in 2014, we intend to pursue a 505(b)(2) regulatory pathway towards NDA approval in the United States. Pursuant to this streamlined development approach, we are permitted to rely, at least in part, on FDA findings of safety and/or effectiveness for a previously approved drug. Based upon written feedback received from the FDA in 2014, we believe this approach will create the opportunity for us to leverage existing data developed with certain existing omega-3 fatty acids to create a streamlined approach to potential approval for MAT9001 for the treatment of severe hypertriglyceridemia (≥500 mg/dL). Simultaneously with those preclinical and clinical studies necessary for approval of this initial indication, we intend to conduct two additional studies designed to highlight the differentiated profile of MAT9001 vs. market leading omega-3 fatty acids and potentially yielding superior data in similar patient populations to those data generated by already-approved omega-3 fatty acids. We believe this dual development strategy will best position MAT9001 to secure an approval to treat severe hypertriglyceridemia as quickly as possible while also positioning MAT9001 as the best-in-class prescription omega-3 therapy as this market and regulatory requirements evolve.
While we advance MAT9001 toward pivotal trials in the cardiovascular space, we are also determined to maximize the value associated with our unique and potentially disruptive lipid nano-crystal (LNC) platform delivery technology. Our proprietary LNC platform delivery technology, licensed from Rutgers University on an exclusive worldwide basis, nano-encapsulates molecules and is designed to render these molecules orally bioavailable, well-tolerated and safe via fusogenic intracellular delivery. We believe the ability of our drug delivery technology to efficiently deliver drugs intracellularly may result in the targeted and safe delivery of pharmaceuticals directly to the site of infection or inflammation as well as the potential to treat a variety of cell-based pathogens, diseases and conditions. We believe our cochleate technology provides us with a highly stable, efficient and broadly applicable drug delivery platform, that has the potential to deliver a broad range of therapies, including small molecules, vaccines, peptides and proteins, as well as nucleic acid polymers in the gene therapy space (e.g., siRNA, mRNA, and CRISPR/Cas-9) in diseases and conditions exhibited by inflammation (e.g., CNS and infectious diseases) as well as intracellular diseases (e.g., intracellular pathogen-related, genetic disorders, and cancer).
Our lead drug candidate based on the LNC platform is MAT2203, an oral formulation of amphotericin B, a well-known and highly-effective, antifungal drug (though traditionally highly-toxic and currently only available in an intravenous formulation) currently used and approved used to treat a variety of invasive, and potentially deadly, fungal infections. MAT2203 has been developed to date with the assistance and financial support of the National Institutes of Allergy and Infectious Disease (NIAID) of the National Institutes of Health (NIH). MAT2203 has been designated as a Qualified Infectious Disease Product (QIDP) with Fast Track Status for the treatment of invasive candidiasis, the treatment of aspergillosis and the prevention of invasive fungal infections in patients who are on immunosuppressive therapy. We have completed two Phase 2 studies of MAT2203 since 2015 and, leading up to and following a meeting with the Office of Antimicrobial Products (OAP) in January 2018, we had been positioning MAT2203 for an initial indication for the prophylaxis, or prevention, of invasive infections in patients who are suffering from acute lymphoblastic leukemia (ALL) who are rendered immunosuppressed due to the therapies being utilized to treat these patients’ leukemia.
While we continue to believe that MAT2203 could become an important solution to the significant unmet medical need to prevent invasive fungal infections in immunosuppressed patients, we believe there are opportunities for a potentially more rapid approval of MAT2203 for the treatment of certain invasive fungal infections in areas of high unmet medical need which can be substantially supported by non-dilutive government funds. In partnership with the NIH, we have conducted numerous preclinical studies of MAT2203 for the treatment of cryptococcal meningitis. In such studies, we observed the potential for MAT2203, utilizing our LNC platform delivery technology, to (a) cross the blood-brain barrier, (b) treat this infection and (c) eliminate the toxicity normally associated with liposomal delivery of amphotericin B intravenously.
This data attracted the attention of several organizations, including the NIH, interested in finding a better treatment option for cryptococcal meningitis. The NIH recently approved and fully funded a grant submission from the University of Minnesota to conduct a clinical study of MAT2203 on patients with cryptococcal meningitis located in Uganda, Africa where this disease is very prevalent among the HIV-positive community. In consultation with the NIH and the University of Minnesota we are finalizing a protocol to commence this study during 2019. We believe that this clinical trial could become a registration-quality trial for an indication for MAT2203 to treat cryptococcal meningitis. We intend to finalize this protocol in the near term and then engage with the FDA to outline a potential pathway for approval of MAT2203 based upon this trial. Given the significant challenge associated with treating cryptococcal meningitis, we believe this indication could position MAT2203 with physicians to become the antifungal of choice for the treatment of all invasive fungal infections given the broad-spectrum nature of amphotericin B. Combined with the oral and targeted delivery and safety profile we believe the LNC platform delivery affords, we believe MAT2203 is well positioned to become a best-in-class antifungal drug. In addition, we believe that a demonstration that MAT2203 can effectively cross the blood-brain barrier in humans in this study could potentially position our LNC platform delivery technology to be utilized with molecules designed to treat diseases of the central nervous system exhibited by inflammation. Developing MAT2203 utilizing non-dilutive, government-sponsored, financing allows us to focus our internal cash resources on MAT9001 while advancing MAT2203 and our LNC platform technology into areas of significant unmet medical need and innovative medicine.
| S-2 |
We have been engaged in discussions with various large, well-established and well-financed biotech and global pharmaceutical companies on potential applications of our LNC platform technology in the gene therapy space. Though early stage, these discussions have been based on existing data utilizing our LNC platform technology with complex nucleic acid polymers such as antisense oligonucleotides, mRNA, siRNA, and DNA plasmids. Our success in nano-encapsulating larger nucleic acids such as DNA plasmids, which can be as large as 11 kilobases in length, has also pushed us toward discussions with the NIH and others about utilizing our LNC platform delivery technology in the CRISPR-Cas9 space.
In July 2018, we announced a research collaboration with the National Institute of Neurological Disorders and Stroke (NINDS), part of the National Institutes of Health (NIH), focused on the development of a novel therapy for the treatment of human immunodeficiency virus (HIV) combining targeted antisense oligonucleotides (ASO) and Matinas’ LNC delivery technology. In January 2019 we announced a research evaluation with a top global pharmaceutical company in which our LNC platform technology would be explored in delivering certain nucleic acid polymers, and we continue to pursue additional strategic collaborations with other interested biotech and pharmaceutical partners.
We believe these early stage, proof-of-concept evaluations could provide a more efficient, less expensive pathway to create numerous strategic verticals in areas of innovative medicine relying upon the development expertise and financial resources of well-established pharmaceutical and biotech companies. Our belief is that data from these evaluations could position us to become a licensor of our LNC platform delivery technology to numerous strategic partners better positioned to absorb the risks and costs of drug development while allowing our company to become a royalty aggregator with the potential to generate upfront license, milestone and royalty payments in order to utilize our LNC delivery platform technology.
Strategy
We are focused on creating value through the streamlined and strategic development of MAT9001 for the treatment of cardiovascular and metabolic conditions and the application of our LNC platform delivery technology to solve complex challenges relating to the delivery of small molecules, gene therapies, proteins/peptides, and vaccines. Key elements of our strategy include:
| ● | Strategically advancing MAT9001 into clinical development toward an initial indication for the treatment of severe hypertriglyceridemia (≥500 mg/dL) (SHTG) with the goal of creating additional data further demonstrating the differentiation of MAT9001 from other prescription omega-3 drugs being used to treat a mixed dyslipidemic patient population in a rapidly emerging and expanding omega-3 market. | |
| ● | Expanding application of our lipid nano-crystal (LNC) delivery platform into the gene therapy space through collaborations with sophisticated and well-resourced biotech and pharmaceutical companies in innovative areas of medicine. | |
| ● | Driving MAT2203 to efficacy data in the treatment of cryptococcal meningitis, an area of significant unmet medical need, with the non-dilutive financial support of the NIH. |
| S-3 |
MAT9001
Our lead cardiovascular product candidate, MAT9001, is a proprietary prescription-only omega-3 fatty acid composition, comprised of a complex mixture of omega-3 fatty acids, including eicosapentaenoic acid, or EPA, docosapentaenoic acid, or DPA, several other omega-3 fatty acids, and relatively nominal amounts of docosahexaenoic acid, or DHA, and non-omega-3 fatty acids. We believe that based upon MAT9001’s unique composition, which includes more DPA than other known omega-3 fatty acids, it will prove to be differentiated from other existing therapies for the treatment of very high triglycerides, or severe hypertriglyceridemia, and dyslipidemia. Triglycerides are fats that are carried in the blood, together with cholesterol, within lipoproteins. High levels of triglyceride rich lipoproteins are associated with an increased risk of atherosclerotic cardiovascular disease and in the case of severe hypertriglyceridemia, acute pancreatitis. High levels of triglycerides are due to both genetic and environmental factors and are associated with comorbid conditions such as diabetes, chronic renal failure and nephrotic syndrome. Unlike the current approved therapies in this product category, many of which have been repurposed following clinical failures in their originally intended indications, we have specifically designed and developed MAT9001 to treat severe hypertriglyceridemia and dyslipidemia. We believe that the results of these targeted development activities and related clinical investigations may yield an improved therapeutic profile compared to the currently-existing therapies, characterized most importantly by MAT9001’s differentiating mechanistic features associated with its unique high DPA composition and enhanced potency as observed in our head to head clinical trial versus Vescepa.
We are primarily focused on developing MAT9001 through approval by the FDA, with an initial indication for the treatment of severe hypertriglyceridemia. Severe hypertriglyceridemia refers to a condition in which patients have high blood levels of triglycerides (TG ≥ 500 mg/dl) and is recognized as an independent risk factor for pancreatitis and cardiovascular disease. If we receive FDA approval for severe hypertriglyceridemia, we may seek approval for use of MAT9001 in additional indications, including the treatment of patients with mixed dyslipidemia who are already undergoing treatment with a statin, a commonly used class of cholesterol lowering medications. Mixed dyslipidemia refers to a condition in which patients have a combination of both elevated triglycerides (≥200mg/dl), and elevated cholesterol levels. According to the NCEP Guidelines, we estimate that approximately 30 to 35 million Americans have mixed dyslipidemia.
Hypertriglyceridemia and Cardiovascular Disease Market Overview
Hypertriglyceridemia refers to a condition in which patients have levels of triglycerides in their blood above 200 mg/dL. Severe hypertriglyceridemia refers to a condition involving levels of triglycerides equal or above 500 mg/dL. Triglycerides (TG) are fats that are carried in the blood, together with cholesterol and lipoproteins. High levels of triglyceride-rich lipoproteins are associated with an increased risk of atherosclerotic cardiovascular disease. Hypertriglyceridemia is due to both genetic and environmental factors. Environmental factors include obesity, sedentary lifestyle, and high caloric diets. Hypertriglyceridemia is also associated with comorbid conditions such as diabetes, chronic renal failure, and nephrotic syndrome.
The prevalence of hypertriglyceridemia is rapidly increasing in the United States and throughout the world, correlating with the increasing incidence of obesity. Severe hypertriglyceridemia is also associated with markedly increased risk for cardiovascular disease and recent studies have demonstrated that elevated triglyceride levels can be regarded as an independent risk factor for cardiovascular disease-related events such as myocardial infarction, ischemic heart disease, and ischemic stroke.
Heart attacks, strokes and other cardiovascular events represent the leading cause of death and disability among men and women in western societies. More than 1 out of every 3 adults in the United States (approximately 92 million) currently lives with one or more types of cardiovascular disease; an estimated 800,000 new or recurrent coronary events and 795,000 new or recurrent strokes occur each year; an estimated 29 million adults ≥20 years of age have high total serum cholesterol levels (≥240 mg/dL), and an estimated 71 million adults ≥20 years of age have borderline high or high low-density lipoprotein (“bad”) cholesterol, or LDL-C, levels (≥130 mg/dL).
| S-4 |
In addition to cholesterol, lipoproteins such as LDL also carry fats in the form of triglycerides. Hypertriglyceridemia refers to a condition in which patients have high levels of triglycerides in the bloodstream and has been reported to be an independent risk factor for cardiovascular disease. Triglyceride levels provide important information as a marker associated with the risk for heart disease and stroke.
Guidelines for the management of very high triglyceride levels (≥500 mg/dL) suggest that reducing triglyceride levels is the primary treatment goal in these patients to reduce the risk of acute pancreatitis. Treating LDL-C remains an important secondary goal. Other important parameters to consider in patients with very high triglycerides include levels of apolipoprotein B (apo B), non-HDL-C, and very low-density lipoprotein cholesterol (VLDL-C).
It is estimated that over 25 million adults in the United States have elevated triglyceride levels ≥200 mg/dL and that more than 50 million adults in the United States have elevated triglyceride levels ≥150 mg/dL. Additionally, approximately 4 million adults in the United States have very high triglyceride levels (≥500 mg/dL).
Mixed dyslipidemia refers to a condition in which patients have a combination of two or more lipid abnormalities including elevated triglycerides, low HDL-C, and/or elevated LDL-C. Both hypertriglyceridemia and mixed dyslipidemia are components of a range of lipid disorders collectively referred to as dyslipidemia. Dyslipidemia has been linked to atherosclerosis, commonly referred to as hardening of the arteries.
Limitations of Current Therapies
Hypertriglyceridemia (HTG) is a prevalent lipid disorder in approximately 25% of the U.S. adult population. Both epidemiological and genetic data have shown associations between HTG and coronary heart disease. Many of those patients are taking statin therapy directed at lowering the risk of cardiovascular disease (CVD) by lowering their LDL-C levels, primarily. Recently, real world administrative database analyses have reported an increased CVD risk as well as direct healthcare costs associated with HTG despite statin therapy and controlled LDL-C compared to those with TG<150 mg/dL.
There is currently no approved prescription omega-3 to lower TG levels in statin-treated patients with mixed dyslipidemia and persistent high (≥200 mg/dL and <500 mg/dL) TG levels due to uncertainty raised by FDA in 2013 regarding the benefit, if any, of drug-induced changes in lipid/lipoprotein parameters beyond statin-lowered LDL-C on cardiovascular risk among statin-treated patients with residually high TG.
Additionally, recent cardiovascular (CV) outcomes trials and meta-analyses with low dose omega-3 fatty acid mixtures containing DHA have not shown substantial benefit in patients receiving contemporary medical therapy, including statins. Due to these failed low dose omega-3 CV outcomes trials, the European regulatory authorities have concluded that omega-3 fatty acid medicines at a dose of 1-gram per day are not effective in preventing further events for patients who have had a heart attack.
TG levels provide important information as a marker associate with the risk for heart disease and stroke, especially when an individual also has low levels of high-density lipoprotein cholesterol (HDL-C) and elevated levels of low-density lipoprotein cholesterol (LDL-C). Multiple epidemiological, clinical, and genetic studies suggest that patients with elevated TG levels (>=200 mg/dL) are at a greater risk of coronary artery disease (CAD) and pancreatitis, a life-threatening condition, as compared to those with normal TG levels. The genes regulating TGs and LDL-C are equally strong predictors of CAD, unlike HDL-C which is not. Other studies suggest that managing and lowering TG levels may reduce these risks.
| S-5 |
Currently Available Treatment Options and Market Opportunity
The dramatic rise in obesity over the last few decades has led to a concomitant increase in cholesterol and triglyceride levels among the population. The collective term for high blood lipid levels such as high cholesterol and high triglyceride levels often used is “dyslipidemia.” Observational studies have resulted in an increased awareness of the critical role that high cholesterol and high triglyceride levels have as a predictor of cardiovascular events. Accordingly, the introduction of new drugs and novel mechanisms of action to lower the risk of cardiovascular events has become a priority. The initial treatment recommendation for patients with dyslipidemia is typically a low-fat diet. If that is not effective, dyslipidemia is then often treated with statins, which account for approximately 80% of all dyslipidemia prescriptions. Statins became a highly successful class of medications for the treatment of dyslipidemia due to their ability to reduce cardiovascular risk in patients at high risk for heart attacks, strokes, and other adverse cardiovascular events. Because of these outcome benefits, the statin utilization rate as compared to the incidence and prevalence of dyslipidemia in the general population, which we refer to as the epidemiology, has risen to almost 40% in the United States. However, the primary activity of statins is in the reduction of LDL-cholesterol levels and they have only modest effects on triglyceride levels. Recognizing that statins alone are not very effective triglyceride lowering drugs, the National Cholesterol Education Program panel recommends the use of more focused therapies to lower triglyceride levels in patients with severe hypertriglyceridemia. Fibrates (a class of amphipathic carboxylic acids), omega-3 fatty acid-based medications and niacin have all been utilized to lower triglycerides levels. In patients with severe hypertriglyceridemia, first-line drug therapy is often a prescription omega-3 or fibrate. Prescription omega-3 based products have been shown to reduce triglyceride levels in the range of 20%-45%.
The treatment rate of hypertriglyceridemia has remained relatively low – below ten percent - compared to the adult population with hypertriglyceridemia. Historically, fibrates such as gemfibrozil (Lopid) and fenofibrate (Tricor or Trilipix) have led the class of treatments of hypertriglyceridemia. However, due to their inability to establish clinical outcome benefits and their limited compatibility with statin therapy, the fibrate utilization rate has remained relatively low and is currently declining. Other products used to treat severe hypertriglyceridemia incorporating niacin as the active pharmaceutical ingredient have not been able to establish additional outcome benefits as compared to statin treatment alone, and are also encountering declining utilization. Because of their lack of outcome benefits, fibrate and niacin use has been mostly concentrated in severe hypertriglyceridemia.
Many omega-3 fatty acid based products have anti-thrombotic and anti-inflammatory effects that suggest effectiveness in inhibiting atherosclerosis in animal models as well as reducing the rate of adverse cardiovascular events in humans at high risk for such events as demonstrated in the JELIS Trial and the GISSI Prevenzione trial in Italy. Furthermore, omega-3 fatty acid based products, either concentrates of both EPA and DHA or EPA alone, have been demonstrated in multiple clinical trials to lower serum concentrations in patients with hypertriglyceridemia. In a recent third-party study, increased levels of EPA and DHA in red blood cells directly correlated with significant reductions in cardiovascular health risks. However, omega-3 fatty acid based medications with significant levels of DHA have been shown to increase LDL-cholesterol levels, which is a negative side effect.
The global prescription omega-3 market has been growing steadily over the last two decades and we estimate the market currently is approaching $2 billion in global sales. The leading omega-3 prescription pharmaceutical products currently approved for the treatment of hypertriglyceridemia are Glaxo Smith Kline’s Lovaza (omega-3-acid ethyl esters, an omega-3 mixture containing mostly EPA and DHA, branded as Omacor in the rest of the world), Omacor and Seacor, very similar to Lovaza and marketed in Europe; and Mochida Pharmaceutical Co., Ltd’s (“Mochida”) Epadel (98% ethyl eicosapentaenoate), the leading Japanese omega-3 product. Recently, a new omega-3 based medication, Amarin’s Vascepa (97% ethyl eicosapentaenoate), was approved and launched in the United States. In addition, Astra Zeneca has an FDA-approved product, Epanova, which has not yet been launched.
| S-6 |
MAT9001 Differentiation Strategy
In contrast to certain other omega-3 based prescription products, MAT9001 is not a product repurposed from a previous development program for another disease or condition, as it was specifically designed for the treatment of severe hypertriglyceridemia and mixed dyslipidemia. Specifically, we are pursuing two avenues of differentiation from existing products, including Vascepa and Lovaza:
| 1. | MAT9001 has unique mechanistic features due to its proprietary composition of omega-3 fatty acids, including DPA, which we believe is a key differentiating omega-3 fatty acid component ( i.e. , a component that is neither EPA nor DHA); and | |
| 2. | MAT9001 is designed to have a highly concentrated potency versus other omega-3 products due to its free fatty acid formulation and potentially improved bioavailability relative to other omega-3 fatty acid pharmaceutical products, as demonstrated in our head to head study against Vascepa. Unlike ethyl-ester omega-3 fatty acid formulations, MAT9001 does not require enzymatic breakdown in the small intestine before it can be adequately absorbed. These enzymes are secreted in the intestine in response to dietary fats. Therefore, ethyl-ester omega-3 fatty acids are not optimally absorbed unless they are taken with a high-fat meal, which is contraindicated in patients with hypertriglyceridemia. Because MAT9001 is less reliant on meal-fat content for optimal absorption, it has significantly greater bioavailability than the ethyl-ester form under the recommended low-fat diet conditions. |
We believe that based upon both publicly available preclinical and human data generated with MAT9001, as well as independent data associated with one of the key omega-3 components contained in MAT9001, our product has the potential to:
| 1. | Better control cholesterol, and may decrease low-density lipoproteins, or LDL, cholesterol levels; | |
| 2. | Demonstrate superior reduction across numerous lipid biomarkers, including triglycerides, VLDL, non-HDL and Apo-C3 levels; and | |
| 3. | Produce certain gene regulatory effects, such as the down regulation of HMG-CoA reductase and PCSK9. |
In addition, MAT9001 contains a much lower concentration of DHA than certain competitive omega-3 products, such as Lovaza or Epanova (products with mixtures of mostly EPA and DHA). As described above, these products reduce triglycerides as the main desired effect but also have the negative side effect of increasing LDL-cholesterol levels. This side effect is observed with the use of Lovaza and Epanova in patients with severe hypertriglyceredemia as well as in patients with mixed dyslipidemia. In contrast, products with very low concentrations of DHA, such as Vascepa, have not shown the increase in LDL-cholesterol levels relative to placebo in either the severe hypertriglyceridemia or mixed dyslipedemia patient populations. Omega-3 products containing low DHA levels have also demonstrated reductions in LDL-cholesterol and non-HDL-cholesterol levels. We believe MAT9001’s unique composition will produce differentiating results in reducing both cholesterol and triglyceride levels. Further, based on our product design, we believe that MAT9001 is well-positioned to become a leading treatment for severe hypertriglyceridemia if approved by the FDA.
MAT9001’s free fatty acid form of omega-3 differentiates it from competitors and we believe this distinction leads to numerous clinical advantages. In a head to head study vs. Vascepa, as detailed further below, improved absorption characteristics and bioavailability were observed for MAT9001 as compared to Vascepa. Our PK/PD head-to-head trial compared the bioavailability of MAT9001 and Vascepa and it was observed that MAT9001’s free fatty acid form was less reliant on meal-fat content for optimal absorption than Vascepa’s ethyl-ester omega-3 form, which requires a high-fat meal for optimal absorption. It was also observed that patients on a low-fat diet exhibited five times higher blood plasma levels of EPA relative to Vascepa. Additional benefits of MAT9001’s improved bioavailability may include once-a-day dosing, reduced pill burden and accompanying heightened patient compliance. All adverse events (AEs) reported in this study (whether related or unrelated to study drugs) were mild or moderate in severity. The most commonly reported AEs judged as possibly related to the study drug were dry skin and rhinorrhea. No serious adverse events (SAEs) were reported during the conduct of this study. The study medications were well-tolerated by patients in this study as well.
| S-7 |
We believe that MAT9001, with its unique ratios of omega-3 free fatty acids, increased plasma concentrations of EPA compared to Vascepa, potential once-a-day dosing convenience, and observed degree of bioavailability and potential to reduce triglyceride levels as observed in our studies to date, is well-positioned to address significant unmet medical need and become a standard of care in the treatment of hypertriglyceridemia. Furthermore, we believe that MAT9001, due to the gene regulatory effects of DPA, in combination with statins, if approved by FDA, could become a standard of care in patients with mixed dyslipidemia with a prescribing and commercial advantage favoring products with a once per day dosing convenience similar to statins.
Development History
We believe we have optimized the manufacturing process for the active pharmaceutical ingredient of MAT9001 and have completed various preclinical studies and one human clinical trial with the MAT9001 active ingredient. We completed the first preclinical studies of MAT9001 in 2013 with others completed during 2014. In 2015, we announced results from our Head-to-Head PK/PD Trial against Vascepa in which we observed statistical superiority in reducing serum triglycerides, total- and non-HDL-cholesterol, apolipoprotein CIII and PCSK9 levels. The study was a pharmacokinetic and pharmacodynamic, open-label crossover study designed to compare the bioavailability and effects of MAT9001 versus Vascepa on serum triglyceride levels. Forty-two patients were treated with 4 grams/day of MAT9001 or Vacsepa for 14 days, followed by a wash-out period and crossed over to the other treatment arm. Study subjects had fasting TG levels of 200-400 mg/dl without lipid altering therapy, or fasting TG levels of 200 to 350 mg/dL if they were on stable-dose statin monotherapy. MAT9001 was observed to significantly reduce PCSK9 in patients. In this trial, MAT9001 achieved greater median percentage reduction in four of six lipid measures, including total cholesterol, when compared to Vascepa:
| ● | MAT9001 significantly reduced median TG levels by 33.2 percent compared to 10.5 percent for Vascepa (p-value <0.001); | |
| ● | MAT9001 significantly reduced median very low-density lipoprotein cholesterol (VLDL-C) levels by 32.5 percent compared to 8.1 percent for Vascepa (p-value <0.001); | |
| ● | MAT9001 significantly reduced median non-HDL-C levels by 8.8 percent compared to 4.6 percent for Vascepa (p-value=0.027); | |
| ● | MAT9001reduced median HDL-C levels by 11.3 percent compared to 11.1 percent for Vascepa (p-value= 0.337); | |
| ● | MAT9001
reduced median LDL-C levels by 2.4 percent compared to 4.3 percent for Vascepa (p-value=0.116); | |
| ● | MAT9001 significantly reduced median total cholesterol levels by 9 percent compared to 6.2 percent for Vascepa (p-value=0.013). |
MAT9001 also outperformed Vascepa in reductions in apolipoproteins (apo) and PCSK9 as compared to baseline:
| ● | MAT9001 reduced median apolipoprotein B levels by 3.8 percent compared to 0.7 percent for Vascepa (p-value=0.058); | |
| ● | MAT9001 significantly reduced median apolipoprotein AI levels by 15.3 percent compared to 10.2 percent for Vascepa (p-value=0.003); | |
| ● | MAT9001 significantly reduced median apolipoprotein CIII levels by 25.5 percent compared to 5 percent for Vascepa (p-value=0.006); and | |
| ● | MAT9001 significantly reduced median PCSK9 levels by 12.3 percent compared to an 8.8 percent increase in PCSK9 levels for Vascepa (p-value <0.001). |
Pre-treatment median values for lipids, triglycerides, apolipoproteins, and PCSK9 levels were measured. Patients were randomized and put on MAT9001 or Vascepa for 14 days. Following the initial treatment period, there was a 5-week washout period, following which patients were put on the other therapy for 14 days. Forty patients completed the trial. MAT9001 met its primary PK endpoint for bioavailability of omega-3 for MAT9001 relative to Vascepa in this study. Statistical analysis demonstrated superiority of MAT9001 over Vascepa for omega-3 bioavailability (baseline adjusted AUC and Cmax, approximately 6-fold higher with MAT9001 on Day 14, with very high statistical significance).
| S-8 |
MAT9001 Development Plan
Following announcement of our head to head study vs. Vascepa in 2015, due primarily to cardiovascular regulatory and commercial market conditions, as well as limited financial resources, we determined to slow down development of MAT9001 until such time as data became available from Amarin’s cardiovascular outcomes trial, REDUCE-IT™.
Following the release of data from the Amarin REDUCE-IT trial, we have re-initiated our development efforts and activities for MAT9001. With the support of a world-class team of key opinion leaders, clinicians and regulatory experts we have designed a development program for MAT9001 designed to (a) potentially streamline our path to approval for MAT9001 in its initial indication to treat severe hypertriglyceridemia and, (b) create additional data both head to head vs. Vascepa and in a mixed dyslipidemic patient population in order to position MAT9001 to become the potential best-in-class omega-3 prescription product for the treatment and prevention of cardiovascular conditions. Our regulatory strategy with FDA will include leveraging a 505(b)(2) registration pathway, consistent with the feedback we received from FDA during 2014. We are in the process of reactivating our IND and expect to complete this activity in the second quarter of 2019.
Pursuant to our development strategy, we intend to advance MAT9001 into a series of clinical trials designed to (a) complete those studies required for approval of an initial indication to treat severe hypertriglyceridemia (≥500mg/dL) and, (b) complete additional trials to demonstrate the differentiation of MAT9001 vs. competitive approved omega-3 products and also create the potential for label enhancement in a mixed dyslipidemic patient population (patients with triglyceride levels 200-499 mg/dL).
During 2019 we intend to initiate and complete the following studies (a) a 28-day comparative bridging toxicology study, and (b)subject to any feedback from FDA, a comparative clinical bioavailability study (36 healthy volunteers) with key endpoints and assessments to include PK parameters (e.g., AUC, Cmax, Tmax, t1/2) for total EPA, DHA and DPA and comparison of PK parameters for MAT9001 after a high fat meal and also fasting vs. fed.
Following completion of these studies, we intend to request an End-of-Phase 2 Meeting with FDA to review the data from the completed studies and to gain a Special Protocol Assessment (SPA) on our pivotal study protocol for the treatment of severe hypertriglyceridemia. During this meeting, we intend to present the design of a Phase 3 registration study in SHTG patients. We anticipate, subject to feedback from FDA, that the study will be a placebo-controlled study with two dose groups of MAT9001: 2 gram and 4 gram/day. We anticipate that approximately 270 patients will be randomized 1:1:1. It is planned that MAT9001 will be dosed either once or twice daily without regard to meals. The primary endpoint of the study is anticipated to be change in TG levels at Week 12.
In addition to the studies required for approval to treat SHTG, we intend to conduct additional trials, including a comparative PK/PD study vs. Vascepa and a second Phase 3 trial of MAT9001 as an add-on to statin therapy in patients with high triglycerides (200-499 mg/dL) at risk for cardiovascular disease. We anticipate that our second head to head study vs. Vascepa will generate topline data during the second half of 2020.
| S-9 |
MAT2203 - Our Lead Product Using our LNC Delivery Platform Technology
We have leveraged our platform lipid nano-crystal (LNC) platform delivery technology to develop two clinical-stage products that we believe have the potential to become best-in-class drug. Our lead LNC platform product candidate, MAT2203, is an orally-administered LNC formulation of a broad spectrum anti-fungal drug called amphotericin B. We previously had been planning to conduct a Phase 2/3 adaptive-design study of MAT2203 in patients with Acute Lymphoblastic Leukemia (ALL) for the prevention of invasive fungal infections (IFI) due to immunosuppressive therapy. While we continue to believe that MAT2203 could become an important solution to the significant unmet medical need to prevent invasive fungal infections in immunosuppressed patients, we believe there are opportunities for a potentially more rapid approval of MAT2203 for the treatment of certain invasive fungal infections in areas of high unmet medical need which can be substantially supported by non-dilutive government funds. In partnership with the NIH, we have conducted numerous preclinical studies of MAT2203 for the treatment of cryptococcal meningitis. In such studies, we observed the potential for MAT2203, utilizing our LNC platform delivery technology, to (a) cross the blood-brain barrier, (b) treat this infection and (c) eliminate the toxicity normally associated with liposomal delivery of amphotericin B intravenously.
We now plan to initially develop MAT2203 for the treatment of cryptococcal meningitis, one of the most frequent and opportunistic infections in Human Immuno-Deficiency Virus (HIV) patients. Given the high morbidity associated with cryptococcal meningitis in HIV patients, the clinical unmet need is globally very high with the global burden estimated at 1 million cases annually. We believe MAT2203, if approved, to have the potential to become the drug of choice for physicians in the treatment of other invasive fungal infections. We plan to leverage the 505(b)(2) regulatory pathway for MAT2203, in part relying upon FDA’s findings ofthe efficacy of amphotericin B, and anticipate meeting with the FDA in the first half of 2019 to discuss our development plans for MAT2203. We also plan to seek accelerated approval for this indication. We are in the final planning stages for a Phase 2 clinical trial, fully funded by the NIH and conducted by the University of Minnesota at their clinic in Uganda. We believe that this study may have the potential to become a pivotal study to support approval of MAT2203 for the treatment of cryptococcal meningitis, and we also plan to submit an application for Orphan Designation and QIDP Designation during the first half of 2019.
Our second clinical stage LNC-based product candidate is MAT2501, an orally administered, cochleate formulation of the broad-spectrum aminoglycoside antibiotic amikacin which may be used to treat different types of multidrug-resistant bacteria, including non-tuberculous mycobacterium infections (NTM), as well as various multidrug-resistant gram negative and intracellular bacterial infections. In May 2017, we completed and announced topline results from a Phase 1 single escalating dose clinical trial of MAT2501 in healthy volunteers in which no serious adverse events were reported and where oral administration of MAT2501 at all tested doses yielded blood levels that were well below the safety levels recommended for injected amikacin, supporting further development of MAT2501 for the treatment of NTM infections. We have decided to temporarily halt clinical development of MAT2501, in order to prioritize and accelerate the development of MAT9001 and MAT2203 and explore utilization of our LNC platform delivery technology in the gene therapy space.
MAT2203 - Product Profile
MAT2203 is an orally-administered, LNC formulation of amphotericin B (a broad-spectrum fungicidal agent). Little to no clinical resistance has been reported to date with amphotericin B as compared to the rapidly emerging drug resistance seen in other antifungal therapies. Currently, IV-only administered amphotericin B is the only broad spectrum fungicidal; however, it has significant treatment-limiting side effects, most notably nephrotoxicity. The ability to provide amphotericin B orally using our proprietary and novel oral formulation comprising our LNC platform delivery technology, may offer a new and promising alternative for patients and doctors. In a clinical Phase 1 single-dose, double-blind, dose-escalating, pharmacokinetic study of 48 healthy volunteers, oral MAT2203 was observed to be well tolerated with no serious adverse events reported, and without any observed nephrotoxicity. The most commonly reported AEs were nausea and abdominal pain. None of the AEs were related to abnormal laboratory evaluations. All treatment emergent adverse events (TEAEs) were mild except 1 instance of “upper respiratory tract infection” which was moderate in a subject following 800 mg MAT2203. No AEs led to withdrawal. There were no serious AEs. There was one pregnancy (subsequently determined that the conception date was 1 to 2 days prior to dosing) resulting in elective termination from the study. More recently, in our Phase 2 trial of MAT2203 conducted by the National Institutes of Health, four out of four enrolled patients met their primary efficacy endpoint, three patients continue on treatment of which two have been successfully taking MAT2203 for more than two years as part of a long term safety extension, with no evidence of kidney or other toxicity frequently associated with the use of amphotericin B.
| S-10 |
Antifungal Market Opportunity
The overall global antifungal market accounted for $10.7 billion in 2015 with estimated annual worldwide sales of prescription systemic antifungal drugs reaching approximately $4 billion. This includes therapies used as active treatment or prophylaxis (preventative) in the inpatient and outpatient setting, therapies used for the treatment of hospitalized patients and therapies used for the treatment of patients who are being discharged from the hospital. We estimate that, each year, there are over 1.5 million cases of invasive fungal infections caused by various species of Candida, Aspergillus and Cryptococcus, the three most common invasive fungal pathogens, globally. The estimated incidence in the U.S. for these conditions is approximately 46,000 for invasive candidiasis, 6,000 for invasive aspergillosis, and 3,000 for cryptococcal meningitis. The rapid progression of disease and high mortality rates (20% - 50%) associated with documented invasive fungal infections often result in antifungal therapy being administered in suspected (unconfirmed) cases or as a preventative measure in patients at high risk. Also, the increasingly widespread use of immune suppressive drugs as cancer chemotherapy or for organ transplantation or treatment of autoimmune disease has resulted in an increasing population of patients at risk for invasive fungal infections. Furthermore, the limited number of systemic antifungal drug classes, consisting of azoles, echinocandins and polyenes, and their extensive use, has led to increased numbers of infections with drug-resistant strains. The Centers for Disease Control and Prevention (CDC) has listed fluconazole-resistant Candida as a serious threat requiring prompt and sustained action and has also identified a rise in echinocandin resistance, especially among Candida glabrata. In June 2016, the CDC issued an extraordinary alert for healthcare facilities and providers to be on the lookout for patients with Candida auris, a multidrug resistant strain with high mortality (approximately 60%). Almost half of C. auris isolates are multidrug resistant to two or more antifungal classes (large majority resistant to fluconazole, 40% resistant to echinocandins). We believe this underscores the urgent need for new agents with demonstrated activity against resistant strains and that can be administered with significantly less toxicity and the potential to discharge patients earlier to reduce hospital stays and associated costs.
Physicians’ options for the treatment of fungal infections are limited by a lack of innovative therapies. Several factors have contributed to the low rate of antifungal drug development, including a previously challenging regulatory environment that necessitated large and costly clinical trials. As a result of this regulatory environment and other factors, the number of antifungals in development has decreased, while anti-microbial resistance has increased.
Our Solution – MAT2203
Our lead anti-infective product candidate, MAT2203, is an application of our LNC platform delivery technology to a broad spectrum anti-fungal drug called amphotericin B. Amphotericin B is an IV administered drug used as a last resort for treatment of systemic fungal infections resistant to triazoles and echinocandins, including resistant candidiasis, cryptococcal meningoencephalitis, and aspergillosis. To date, there have been little to no reports of clinically observed drug-resistance to amphotericin B, further bolstering the use of this compound as the most likely last resort treatment for fungal infections in the foreseeable future. However, the use of amphotericin B is relatively limited because it is currently only available as an IV-administered product and has documented history of severe toxicity (most notably nephrotoxicity). By utilizing our LNC platform delivery technology to nano-encapsulate amphotericin B, there is now an opportunity for the drug to be administered orally with targeted delivery to infected cells, which we believe may have fewer side effects than the currently available IV-formulations of amphotericin B. Our LNC delivery of amphotericin B changes the bio-distribution, resulting in a higher level of the drug at the site of infection and a lower level of circulating amphotericin B. By reducing the amount of circulating drug, our LNC may reduce overall toxicity. Importantly, drug concentrations will be high only in tissues due to the migratory nature of macrophages to inflammatory regions. Based upon our studies to date, we believe MAT2203 has the potential to offer improved safety and reduced toxicity and, as a result, we believe MAT2203 will be able to offer a categorically different formulation that delivers orally administered amphotericin B, directly to the target cell at the site of infection. In collaboration with the NIH, in multiple studies, we have demonstrated in cryptococcal meningitis mouse models that our LNC-delivered amphotericin b, following oral administration, has the ability to successfully cross the blood brain barrier to the site of infection in mice. This demonstration provides important data indicating that our LNC platform delivery technology could become an important delivery solution for a variety of CNS-based disorders and diseases.
| S-11 |
We believe that MAT2203 has the potential to become a best-in-class induction, consolidation, and maintenance therapy for the treatment of cryptococcal meningitis in HIV patients by offering the following key benefits:
| ● | Potential to treat resistant pathogens. We believe that MAT2203 has the potential to prevent and treat fungal infections caused by drug resistant fungi, including those resistant to existing azoles and echinocandins, due to amphotericin B’s fungicidal (i.e. killing the fungi) nature and potency against resistant strains and the potential for our cochleate drug delivery platform to provide higher drug exposure early in the course of therapy. | |
| ● | Enabling an all-oral therapy. Cryptococcal meningitis has become the most common cause of adult meningitis in many parts of Africa, where cryptococcosis now rivals tuberculosis in all-case mortality. While long-term survival has improved with widespread use of antiretroviral therapy in high income countries, early mortality remains high. Early mortality rates are often ~ 70% in routine practice where access to diagnostics or medications is limited or unavailable, intracranial pressure is uncontrolled, or in settings where other barriers to the management of cryptococcal meningitis exist. IV administration of amphotericin B deoxycholate is not often possible in resource-limited settings, even when it is available. | |
| ● | Shorter and less costly hospital stays and lower outpatient costs. By providing physicians and patients with access to an orally available, broad spectrum fungicidal agent in MAT2203, there is the potential to reduce hospital costs, which account for over 70% of the overall treatment cost of invasive fungal infections. |
The FDA has granted MAT2203 designations for Qualified Infectious Disease Product, or QIDP, and Fast Track for the treatment of invasive candidiasis and aspergillosis and for the prevention of IFIs in patients on immunosuppressive therapy. We are in the process of applying for a fourth QIDP for the treatment of cryptococcal meningitis. We will also apply for Orphan Drug Designation for MAT2203 for the treatment of cryptococcal meningitis. The FDA may designate a product candidate as an orphan drug if it is intended to treat a rare disease or condition, which is generally defined as having a patient population of fewer than 200,000 individuals in the United States, or a patient population greater than 200,000 in the United States where there is no reasonable expectation that the cost of developing the drug will be recovered from sales in the United States. The orphan drug designation provides eligibility for orphan drug exclusivity in the United States upon FDA approval if a product that has orphan drug designation subsequently receives the first FDA approval for a particular active ingredient for the disease for which it has such designation. For a product that obtains orphan drug designation on the basis of a plausible hypothesis that it is clinically superior to the same drug that is already approved for the same indication, in order to obtain orphan drug exclusivity upon approval, clinical superiority of such product to this same drug that is already approved for the same orphan indication must be demonstrated. Orphan drug exclusivity means that the FDA may not approved any other applications, including an NDA, to market the same drug for the same indication for seven years, except in limited circumstances such as if the FDA finds that the holder of the orphan drug exclusivity has not shown that it can assure the availability of sufficient quantities of the orphan drug to meet the needs of patients with the disease or condition for which the drug was designated. Similarly, the FDA can subsequently approve a drug with the same active moiety for the same condition during the exclusivity period if the FDA concludes that the later drug is clinically superior, meaning the later drug is safer, more effective or makes a major contribution to patient care. Orphan drug designation also entitles a party to financial incentives such as opportunities for grant funding towards clinical trial costs, a waiver from payment of user fees, an exemption from performing clinical studies in pediatric patients unless the FDA requires otherwise by regulation, and tax credits for the cost of the clinical research. The QIDP designation, provided under the Generating Antibiotic Incentives Now Act, or the GAIN Act, offers certain incentives for the development of new antibacterial or antifungal drugs, including eligibility for Fast Track designation, priority review and, if approved by the FDA, eligibility for an additional five years of marketing exclusivity. Fast Track designation enables more frequent interactions with FDA to expedite drug development and review. Fast Track designation does not change the standards for approval and we can provide no assurances that we can maintain Fast Track designation for MAT2203 or that such designation will result in faster regulatory review. The seven-year period of marketing exclusivity provided through orphan designation, if granted, combined with an additional five years of marketing exclusivity provided by the QIDP designation positions MAT2203 with a potential for a total of 12 years of marketing exclusivity to be granted at the time of FDA approval. Our plan is to further secure QIDP/Fast Track/Orphan Designation for the initial development target indication of cryptococcal meningitis.
| S-12 |
Development History of MAT2203 and Initial Target Indication
MAT2203 was studied in animal model studies of various fungal infections including invasive candidiasis, aspergillosis and cryptococcal meningitis.
The data from animal studies for MAT2203 indicate a side-effect advantage over other amphotericin B formulations, which we believe is based on two phenomena:
| ● | The lipid-crystal nano-particle is a solid particle that does not significantly “leak” its drug content while circulating. The particle releases its medication pay-load only when inside the target cells, and thus appears that the use of MAT2203 does not result in toxicities normally seen in the kidneys when using current formulations of amphotericin B. | |
| ● | Because of this targeted approach, we have been able to increase the therapeutic window on a mg/kg basis as compared to IV amphotericin B formulations. We have observed equivalent efficacy at lower doses as well as been able to use oral doses of up to 10x the highest tolerable IV dose in animal model studies. |
NIH-Conducted Study
In early 2017, we reported interim data from the NIH-Conducted Phase 2a Clinical Study of Orally-Administered MAT2203 for the Treatment of Chronic Refractory Mucocutaneous Candidiasis. At that time, two out of the two patients with long-standing azole resistant mucocutaneous candidiasis met the primary endpoint of the Phase 2a study, achieving ≥ 50% clinical response with treatment of MAT2203. Patient #01 achieved a 57% reduction in clinical symptoms after 8 weeks on therapy while patient #02 achieved an 85% reduction in such clinical symptoms after 6 weeks of treatment. MAT2203 was well tolerated with majority of adverse events observed being mild in severity and mostly unrelated to study drug. Importantly, for both patients renal and liver function parameters remained well within normal ranges during the core study as well as during the first 6-month extension of this study. In July 2017, the NIH/NIAID institutional review board approved continuation of treatment of patients in the study-extension for an additional 6 months, for total extension of up to one year.
In January 2018, the National Institutes of Health (“NIH”) reported positive data from a third patient enrolled in this study. This third patient, with long-standing azole resistant mucocutaneous candidiasis, met the primary endpoint of the Phase 2a study in achieving ≥ 50% clinical response with treatment of MAT2203. MAT2203 was well tolerated with any adverse events observed being mild in severity and unrelated to study drug. With this third positive response, the study has met its statistical hurdle for success. In June of 2018, the NIH reported that a fourth patient had enrolled in the study and had met the primary endpoint in achieving ≥ 50% clinical response with treatment of MAT2203. All four patients had been enrolled in a long-term study extension and the initial two patients have now shown no signs of kidney or liver toxicity over the approximately twenty-four months of being administered MAT2203. The third patient was required to drop out of the long-term safety portion of the study due to the development of an infection that does not respond to amphotericin B. The fourth patient continues in the long-term safety extension for the study. The clinical response to MAT2203 seen in all three patients continuing on drug has been maintained and/or improved during the extension period in addition to patients reporting meaningful quality-of-life improvements.
VVC Study
In late 2017, we announced the topline data from our Phase 2 study in Vulvovaginal Candidiasis (VVC) using MAT2203. In the context of our overall program for MAT2203 with the aim to develop our lead product initially for the prevention of invasive fungal infections in patients who are immunocompromised due to immunosuppressive therapy, our goal was, in addition to further establishing the safety and tolerability of MAT2203, to demonstrate efficacy of MAT2203 through a mechanism involving systemic absorption in a non-life threatening fungal infection. This study concept was consistent with early human efficacy studies in the development of other anti- fungal therapies. This Phase 2 study was not designed or powered to support an indication for the treatment of VVC and therefore supplant fluconazole as the standard of care. The key data generated from this study included additional safety and tolerability data.
| S-13 |
In this VVC study, the primary endpoint of safety was met, and it was demonstrated that oral delivery of encochleated amphotericin B is safe and well tolerated without the renal and hepatic toxicities that can be seen with administration of intravenous amphotericin B. Drug-related treatment emergent adverse events in this study were mostly of mild and gastro-intestinal nature and were seen at a rate of 20%, 18% and 2% respectively for MAT2203 200mg, MAT2203 400mg, and fluconazole. Consistent with the safety observations in the NIH study, in this VVC study no drug-related effects on liver function were observed and kidney function parameters stayed within normal ranges during the entire study for all 91 patients treated with MAT2203 for 5 days.
Development Plan
In February 2019, the NIH approved the funding of a planned clinical trial related to a grant application submitted by The University of Minnesota to study MAT2203 for the treatment of cryptococcal meningitis. The clinical study will be conducted in Uganda and has already received the approval of all necessary regulatory authorities. The protocol is in the process of being finalized and the trial is anticipated to consist of two parts with the following design. The initial portion of the trial will be a Phase 1 Study conducted in HIV patients in Uganda without active neurological infection. Subjects in this initial phase of the study will involve a determination of the maximal tolerated dose to arrive at an optimal dose for the second part of the study. A Data Monitoring Committee (DMC) will review the data from the initial phase prior to commencing the efficacy portion of the trial. Upon review of the data, the DMC will make a recommendation to the Ethics Committee/IRB as to the dose to be used in the efficacy portion of the study in patients with active neurological infection. The efficacy portion of the study has been designed as an open-label trial to evaluate the safety, tolerability, and microbiologic efficacy of oral MAT2203 as part of induction and consolidation treatment of HIV-infected patients with cryptococcal meningitis compared with standard intravenously delivered amphotericin B. Participants in the study will be enrolled in sequential cohorts designed to mitigate the risk associated with these very sick patients. Induction treatment in each cohort will start with IV amphoterticin and flucytosine treatment with oral MAT2203 administered as step-down treatment for the initial two cohorts (with earlier step-down to MAT2203 in each subsequent cohort.) The next two cohorts will test the induction of treatment with our MAT2203 product, with step-down treatment to IV amphotericin. The final cohort of patients will have a MAT2203 induction treatment (plus flucytosine) without IV administered amphotericin b. The primary endpoint for this trial will be the rate of cerebrospinal fluid (CSF) Cryptococcus clearance as measured by serial quantitative CSF fungal cultures. This study is planned to commence in the second half of 2019. The trial design is expected to be finalized during 2019 following a meeting with FDA.
We are currently completing a 90-day rat toxicology study to support dosing with MAT2203 beyond the current 28-day tox coverage. The in-life portion of the study has been completed. There have been no signs of toxicity noted to date.
Our plan is to meet with FDA to review our development plan and study design as we intend to conduct this Phase 2 study in patients with cryptococcal meningitis under a US IND. We additionally will discuss with FDA our plans to leverage a 505(b)(2) Pathway, relying, in part, upon FDA’s findings of safety and efficacy of I.V. amphotericin B.
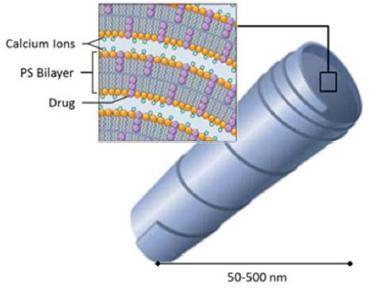
Our Cochleate Platform Delivery Technology
Cochleate lipid-crystal nano-particles are composed of simple, naturally occurring materials: phosphatidylserine (PS) and calcium. They are stable and have a unique multilayered structure consisting of a large, continuous, solid, lipid bilayer sheet rolled up in a spiral or as stacked sheets, with no internal aqueous space (Figure 1). This unique structure provides protection from degradation for “encochleated” molecules. Components within the interior of the cochleate remain intact, even though the outer layers of the cochleate may be exposed to harsh environmental conditions or enzymes.
| S-14 |
Figure 1 Cochleate Formulation
The structure is formed when a series of solid lipid sheets engulf drug molecules, a process referred to as “encochleation.” Encochleation, developed by Matinas and Rutgers New Jersey Medical School, involves combining calcium and soy-derived PS, two naturally occurring materials classified as GRAS (generally recognized as safe), through a stirring process to envelop the active pharmacological ingredient. The result is a nano-size encochleated drug formulation (Figure 2).
Figure 2 Formation of Cochleate
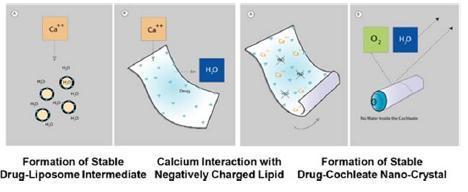
Cochleates have been shown to improve existing drugs by providing 1) cell-targeted delivery; 2) reduced blood levels thereby reducing toxicity; and 3) oral delivery of drugs now only available intravenously. Cochleates work by encapsulating molecules of drugs in a solid, anhydrous, crystalline structure, protecting them as they pass through the gastrointestinal (GI) tract where they cross the mucous membrane. Once the cochleates have crossed the mucosal barrier of the GI tract into the lymphatic system, they are picked up by particle scavenging cells of the mononuclear phagocytic system, such as macrophages and dendritic cells. (Figure 3). Activated macrophages, with drug-cochleate inside, migrate to the site of infection or to the target organ and deliver amphotericin B.
| S-15 |
Cells in the mononuclear phagocytic system are immune cells that have the capacity to engulf and destroy numerous potentially pathogenic materials and organisms within the body. These cells are found in almost every site of the body, save a few ‘immune privileged’ sites (e.g. eyes, fetus, and testes). Such cells help with non-specific (innate) immune defenses as well as help initiate specific (adaptive) immune responses, thus they play a critical role bridging the gap between innate and adaptive immune responses. Our core capabilities combine the use of lipids as active pharmaceutical ingredients (API) and the use of lipids in “cochleate-shaped” lipid-crystal nano-particle drug delivery vehicles. Therapeutic applications of our proprietary delivery technology were initially focused on the delivery of several potent and highly efficacious anti-fungal and anti-bacterial agents which are currently still associated with serious side effects, including irreversible toxic effects on kidney and hearing function. We believe our technology has the potential for targeted delivery of these agents, which positions us to be at the forefront of dealing with these very serious problems. We have now also expanded our research and development efforts for our LNC Platform to focus on the delivery of a wide range of therapeutic treatments, in particular those in the oligonucleotide space (siRNA, DNA, antisense DNA, mRNA, and CRISPR-Cas9). We continue to push forward our business development efforts to further expand our collaborations across pharma and biotech companies who have innovative therapies with delivery challenges which may be addressable with our LNC platform delivery technology.
Our LNC technology is currently being used to encapsulate potent anti-infective drugs in tiny lipid-crystals which are selectively picked up by cells in the mononuclear phagocytic system, such as macrophages, and transported to infected cells. These tiny lipid crystals are referred to as “cochleates.” Cochleates have a multilayer crystalline, spiral structure with no internal aqueous space. The structure is formed when a series of solid lipid sheets roll up and engulf drug molecules in between the sheets, a proprietary process referred to as “encochleation”. The result is a lipid-crystal encochleated drug formulation made up of nano-sized particles. We believe our cochleate delivery technology provides an effective delivery mechanism without chemically bonding or otherwise altering the drug. Because the medications are locked in the particles, we believe the exposure to sensitive organs will be reduced, potentially resulting in reduced toxic effects. In summary, we believe this unique technology offers (1) targeted delivery, (2) decreased toxic effects, and (3) oral formulation (even for IV-only medications).
Multi-organ Protection: The key innovation of our cochleate delivery technology is our ability to package medication inside lipid-crystal particles without leaking. Because of their crystal nature, these particles are truly solid and hold on tightly to their medication pay-load. This is where the cochleate delivery technology differs markedly from other lipid-based delivery technology, such as liposomal delivery. Liposomes are liquid delivery systems which typically leak some of their drug content into the circulatory system, thus still exposing vulnerable organs and tissues to potential toxic effects. Keeping potentially organ-toxic medications inside the lipid-crystal particles strongly differentiates our cochleate delivery technology from other drug-delivery approaches.
Targeted Delivery: The size of our individual cochleate lipid-crystals is typically in the range of 50-500 nm. This is very small and by comparison close to the size of a large virus or a small bacteria. Our body produces several cell-types that are designed to remove viruses and bacteria from our system. These cell types, such as macrophages, are part of our immune system and “swallow” the bacteria and viruses they encounter in order to protect us from infections. Because of the size our lipid-crystal cochleate particles and the phospholipid surface structure (the cell membranes of bacteria are also made up from phospholipids), macrophages tend to absorb these cochleate particles very well.
Oral Formulation: Many drugs that are currently on the market are only effective in treating diseases when administered via IV. For example, many anti-infective drugs must be administered via IV in order to be effective. IV administration presents several challenges to care, such as risk of infection, patient discomfort from injections, and higher cost of care than anti-infective drugs that can be taken orally (IV delivery must be performed by a doctor or nurse, often within a very expensive hospital setting). Although several technologies have been used to attempt to convert IV drugs to orally delivered medications, success has been limited due to the difficulty in achieving adequate bioavailability (i.e., the amount of drug that is absorbed into the body) with oral formulation. We believe that the unique cochleate crystal-structure in our platform technology protects the drug from degradation when it passes through the gastrointestinal (GI) tract and that its lipid surface features facilitate the particle to be absorbed into the blood stream. The potential application of our cochleate delivery technology for the delivery of injectable medications offers significant clinical and commercial value if successfully demonstrated in human clinical trials. It is our intent to further validate the LNC Platform technology in our planned cryptococcal meningitis study.
| S-16 |
Our cochleate lipid-crystal nano-particle technology changes the delivery of medicines in a unique manner and alters the bio-distribution of these medications by targeting tissues and organs that are affected by infection and inflammation. Besides IV-only anti-infectives such as amphotericin B and amikacin, we have orally delivered in animal studies the influenza vaccine, siRNA, NSAIDs, other anti-infectives such as atovaquone, and many other compounds across multiple therapeutic areas, demonstrating the potential broad application of our technology. We have observed rapid local accumulation in infected tissues, which appear be the result of transport of our drug-loaded cochleates by macrophages and other immune-cells. For example, in a mouse model of invasive candidiasis, comparing orally administered MAT2203 to injected amphotericin B deoxycholate (original drug Fungizone), we observed amphotericin B levels above the minimal inhibitory concentration inside infected organs on day 1 with MAT2203 treatment while such levels were not reached with the injected original amphotericin-deoxycholate product until 3-4 days of treatment. Such kinetics have been seen before with other medications, such as macrolide antibiotics (e.g. azithromycin). It appears from our data that the kinetics of cochleate delivery has similarities to the kinetics of macrolide antibiotics. We expect that additional preclinical and clinical work on the kinetics of our cochleate products will further elucidate the mechanism of cochleate delivery to the site of infection or inflammation.
Strategic Collaborations Using LNC Technology
We believe our LNC platform delivery technology can be used to reformulate a wide variety of molecules and drugs which, (i) require delivery technology to effectively protect molecules and drugs in the body and could benefit from efficient delivery and cellular uptake by target cells, and (ii) are currently only available in IV formulations or, (iii) otherwise experience significant toxicity-related adverse events. Leveraging our cochleate delivery technology, we believe we can develop a robust pipeline of product candidates, either internally or through robust strategic partnerships with pharmaceutical and biotech companies. We have tested a range of pharmaceutical compounds reformulated by our cochleate delivery technology in proof-of-concept animal studies, including oligonucleotides (mRNA, siRNA, DNA plasmids), vaccines, anti-inflammatory agents, NSAIDs and atovaquone. By way of example, in 2016 we received a patent issuance related to LNC compositions directed against expressions of proteins. The allowed patent claims cover our proprietary methods related to the composition and the formation of encochleated siRNA for potential use as therapy for regulating gene expression. We intend to pursue opportunities to develop products, either alone or in partnership with other pharmaceutical or biotech companies, related to this technology and this remains a key part of our strategy to maximize the value of this unique and disruptive lipid-crystal nanoparticle delivery technology.
We continue to actively collaborate with the NIH on a number of therapeutic fronts to further expand the generation of data to support broad use of our LNC platform technology across broad therapeutic treatment modalities. In July 2018, we announced a research collaboration with the National Institute of Neurological Disorders and Stroke (NINDS), part of the National Institutes of Health (NIH) focused on the development of a novel therapy for the treatment of human immunodeficiency virus (HIV) combining targeted antisense oligonucleotides (ASO) and our LNC delivery technology. The goal of this collaboration is to leverage the unique attributes of our LNC technology to safely, effectively and efficiently deliver ASO intracellularly to inhibit Trans-Activator of Transcription (Tat)/viral mRNA translation. Tat is a contributing factor in three major aspects of HIV infection post treatment with antiretroviral therapy (ART): viral replication/latency, chronic inflammation and neurological complications. Tat is a key regulatory protein not specifically targeted by currently available ART. In vitro and in vivo studies will be conducted to determine optimal structures for incorporating ASOs into the LNC technology platform, delivery into target cells and the effective inhibition of Tat and/or viral replication while monitoring Tat-induced cytotoxicity.
In January 2019 we announced a research evaluation with an undisclosed top global pharmaceutical company aimed to evaluate synergistic effects of our lipid-nano-crystal (“LNC”) platform delivery technology with our partner’s nucleic acid polymer technology. Formulations will be developed using our LNC delivery technology which enables the development of a wide range of difficult-to-deliver molecules. Promising formulations will be tested in in vitro and in vivo preclinical studies. For competitive reasons, the agreement stipulates certain confidential provisions, including the pharmaceutical company’s identity, the therapeutic molecule(s), the intended targets and the financial terms of the agreement.
| S-17 |
Exclusive License Agreement with Rutgers University
Through our acquisition of Aquarius Biotechnologies Inc., we acquired a license from Rutgers University for the cochleate delivery technology. The Amended and Restated Exclusive License Agreement between Aquarius and Rutgers, The State University of New Jersey (successor in interest to the University of Medicine and Dentistry of New Jersey) provides for, among other things, (1) a license issue fee of $25,000 paid upon execution, (2) an increased equity interest in the company from 5% to 7.5% of Aquarius (prior to our acquisition of Aquarius in the Aquarius Merger), (3) royalties on a tiered basis between low single digits and the mid-single digits of net sales of products using such licensed technology, (4) a one-time sales milestone fee of $100,000 when and if sales of products using the licensed technology reach the specified sales threshold and (5) an annual license fee of initially $10,000, increasing to $50,000 over the term of the license agreement. We also agreed to assume the responsibility to pay required patent prosecution and maintenance fees covering the technology.
Unless otherwise terminated by either party, the term of the license, on a country by country basis, shall be the longer of 7-1/2 years from the date of first commercial sale of a product in a country using the licensed technology or until the expiration of the last-to-expire patent rights licensed under the agreement, whichever is longer. Rutgers has the right to terminate the license agreement if we have not commenced commercial sales of at least one product using the licensed technology within nine years of the effective date of the license agreement.
Intellectual Property
The proprietary nature of, and protection for, our product candidates and our discovery programs, processes and know-how are important to our business. We will seek to protect our products and associated technologies for their manufacturing and development through a combination of patents, trade secrets, proprietary know-how, FDA exclusivity and contractual restrictions on disclosure. Our policy is to pursue, maintain and defend patent rights and to protect the technology, inventions and improvements that are commercially important to the development of our business. Our success will significantly depend on our ability to obtain and maintain patent and other proprietary protection for commercially important technology and inventions and know-how related to our business, defend and enforce our patents, preserve the confidentiality of our trade secrets and operate without infringing the valid and enforceable patents and proprietary rights of third parties. We also rely heavily on know-how and continuing technological innovation to develop and maintain our proprietary position.
Matinas-Owned Intellectual Property Relating to MAT9001
We have sought patent protection in the United States and internationally for our MAT9001 discovery program, and any other inventions to which we have rights, where available and when appropriate. Our policy is to pursue, maintain and defend patent rights, whether developed internally or licensed from third parties, and to protect the technology, inventions and improvements that are commercially important to the development of our business.
Our current patent portfolio relating to MAT9001 and MAT8800 is comprised of two issued patent U.S. patents and one issued foreign patent in Australia. The issued patents claim cover the Company’s proprietary methods relating to triglyceride levels, total cholesterol, VLDL-cholesterol or apolipoprotein C-III by administering a pharmaceutical composition comprising omega-3 fatty acids including eicosapentaenoic acid (EPA) and docosapentaenoic acid (DPA). These patents provide important protection to MAT9001 through 2033. In addition, we have nineteen additional patent applications across four patent families covering the oil composition for MAT9001, other omega-3 fatty acid compositions, as well as formulations of MAT9001 and similar formulations. All of these filed patent applications also comprise methods of use of such oil compositions and formulations. Any patents that may issue from these filed United States patent applications and their counterpart international application covering the MAT9001 drug substance, formulation, and methods for use in treatment would extend protection until at least 2033.
Exclusively Licensed and Matinas-Owned Intellectual Property Relating to Our Proprietary Cochleate Delivery Technology Platform and MAT2203
The patents and patent applications that we exclusively license from Rutgers University provide patent protection for the proprietary chemistry technology used in our process to make our lipid nano-crystal and geodate cochleates and formulate the active pharmaceutical ingredients delivered inside this delivery technology, as in MAT2203, our lead product comprising the LNC platform delivery technology. Pursuant to our license agreement, we acquired rights to a portfolio that currently includes 11 pending applications and 22 issued U.S. and foreign patents, including 15 patents issued within the last 3 years, which extends patent protection until at least 2033. In addition, we have 28 Matinas-owned pending patent applications filed both in the United States and in foreign jurisdictions within the past 3 years. We have chosen to file these patent applications in selected foreign markets that we consider important for our product candidates. These international markets generally include Europe, China, India, Brazil, Russia, Canada, Japan, Korea, Australia and Mexico. These pending patent applications can extend patent protection through 2037. This patent portfolio covers our cochleate delivery system which covers a broad spectrum of technology, including amphotericin B cochleates, geodate cochleates, methods of delivering nutrients or biologically relevant molecules to a host using cochleates, cochleate vaccine compositions and protein-lipid vesicles, small interfering RNA cochleates, mRNA cochleates methods of enhancing the encochleation of hydrophilic molecules and cochleates made with low purity soy phosphatidylserine.
| S-18 |
We cannot be sure that patents will be granted with respect to any of our pending patent applications or with respect to any patent applications we may own or license in the future, nor can we be sure that any of our existing patents or any patents we may own or license in the future will be useful in protecting our technology. For this and more comprehensive risks related to our intellectual property, please see “Risk Factors—Risks Relating to Our Intellectual Property and Regulatory Exclusivity.”
In addition to patents, we rely on trade secrets and know-how to develop and maintain our competitive position. For example, significant aspects of our proprietary LNC technology platform as well as the manufacture of certain intermediates utilized in MAT9001, as well as our soft gelatin capsule formulation, are based on unpatented trade secrets and know-how. Trade secrets and know-how can be difficult to protect. We seek to protect our proprietary technology and processes, in part, by confidentiality agreements and invention assignment agreements with our employees, consultants, scientific advisors, contractors and commercial partners. These agreements are designed to protect our proprietary information and, in the case of the invention assignment agreements, to grant us ownership of technologies that are developed through a relationship with a third party. We also seek to preserve the integrity and confidentiality of our data and trade secrets by maintaining physical security of our premises and physical and electronic security of our information technology systems. While we have confidence in these individuals, organizations and systems, agreements or security measures may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors. To the extent that our contractors use intellectual property owned by others in their work for us, disputes may arise as to the rights in related or resulting know-how and inventions.
We also plan to seek trademark protection in the United States and outside of the United States where available and when appropriate. We intend to use these registered marks in connection with our pharmaceutical research and development as well as our product candidates.
Competition
The biotechnology and pharmaceutical industries are characterized by rapidly advancing technologies, intense competition and a strong emphasis on proprietary products. We face competition from many different sources, including commercial pharmaceutical and biotechnology enterprises, academic institutions, government agencies and private and public research institutions. Many of these companies have far greater human and financial resources and may have product candidates in more advanced stages of development and many will reach the market before our product candidates. Competitors may also develop products that are more effective, safer or less expensive or that have better tolerability or convenience.
MAT9001
Our competitors both in the United States and abroad include large, well-established pharmaceutical and generic companies, specialty and generic pharmaceutical sales and marketing companies, and specialized cardiovascular treatment companies. GlaxoSmithKline plc currently sells Lovaza®, a prescription-only omega-3 fatty acid indicated for patients with severe hypertriglyceridemia, which was approved by FDA in 2004 and has been on the market in the United States since 2005. Multiple generic versions of Lovaza are available in the United States. Other large companies with competitive products include AbbVie, Inc., which currently sells Tricor® and Trilipix® for the treatment of severe hypertriglyceridemia and Niaspan®, which is primarily used to raise high-density lipoprotein cholesterol, or HDL-C, but is also used to lower triglycerides. Multiple generic versions of Tricor, Trilipix and Niaspan are also available in the United States.In 2012, Amarin Corporation received an approval to market its prescription-only omega-3 ethyl ester called Vascepa® for the treatment of severe hypertriglyceridemia.
| S-19 |
In addition, in May 2014, Epanova® (omega-3-carboxylic acids) capsules, a free fatty acid form of omega-3 (comprised of 55% EPA and 20% DHA), was approved by the FDA for patients with severe hypertriglyceridemia. Epanova was developed by Omthera Pharmaceuticals, Inc., and is now owned by AstraZeneca Pharmaceuticals LP (AstraZeneca). Also, in April 2014, Omtryg, another omega-3-acid fatty acid composition developed by Trygg Pharma AS, received FDA approval for severe hypertriglyceridemia. Neither Epanova nor Omtryg have been commercially launched, but could launch at any time. Each of these competitors, other than Trygg, has greater resources than we do, including financial, product development, marketing, personnel and other resources.
We are also aware of other pharmaceutical companies that are developing products that, if successfully developed, approved and marketed, would compete with MAT9001. Acasti Pharma, or Acasti, a subsidiary of Neptune Technologies & Bioresources Inc., announced in December 2015 that it intends to pursue a regulatory pathway under section 505(b)(2) of the Federal Food, Drug, and Cosmetic Act, or FDCA, for its omega-3 prescription drug candidate, CaPre® (omega-3 phospholipid), derived from krill oil, for the treatment of hypertriglyceridemia.
Akcea Therapeutics/Ionis Pharmaceuticals (formerly Isis Pharmaceuticals), or Akcea/Ionis, in August 2018 announced the receipt of a complete response letter from the FDA for WAYLIVRA™ (volanesorsen), a drug candidate administered through weekly subcutaneous injections, for the treatment of familial chylomicronemia syndrome (FCS). Akcea will continue to work with the FDA on the path forward for Waylivra for the treatment of FCS. Waylivra continues to be developed for the treatment of familial partial lipodystrophy (FPL).
In June 2018, Gemphire Therapeutics announced positive topline results from a Phase 2b trial (INDIGO-1) of its drug candidate, gemcabene, in patients with severe hypertriglyceridemia. Gemcabene is an oral, once-daily pill for a number of hypercholesterolemic populations and severe hypertriglyceridemia.
Zydus Cadila has a Phase 2 development program for its lead molecule, Saroglitazar, in various indications, including severe hypertriglyceridemia in the United States. In August 2018, the Company announced that it had suspended the Phase 2 trial in the severe hypertriglyceridemia indication due to study enrollment issues, while it continues development activities in other indications. The product is approved in India under the name Lipaglyn® for the treatment of hypertriglyceridemia and diabetic dyslipidemia. We are also aware that bezafibrate has been licensed by Intercept Pharmaceuticals to be further developed and potentially launched in the United States market.
MAT2203
Although we believe that our proprietary LNC platform delivery technology, experience and knowledge in our areas of focus provide us with competitive advantages, these potential competitors could reduce our commercial opportunities. For many of our product candidates, we anticipate facing competition from other products that are available on a generic basis and offered at low prices. Many of these generic products have been marketed by third parties for many years and are well accepted by physicians, patients and payers.
We believe that MAT2203 and any other development candidate we may pursue in the future using our proprietary cochleate drug delivery technology platform, paralleled with our scientific and development expertise in the field of drug delivery, provide us with competitive advantages over our peers. However, we face potential competition from various sources, including larger and better-funded pharmaceutical, specialty pharmaceutical, and biotechnology companies, as well as from generic drug manufacturers, academic institutions, governmental agencies and public and private research institutions.
MAT2203 will primarily compete with antifungal classes approved for the treatment of candidemia and mold infections, which include polyenes, azoles and echinocandins. The approved branded therapies for these indications include Cancidas (caspofungin, marketed by Merck & Co.), Eraxis (anidulafungin, marketed by Pfizer, Inc.), Mycamine (micafungin, marketed by Astellas Pharma US, Inc.), Diflucan (fluconazole, marketed by Pfizer, Inc.), Noxafil (posaconazole, marketed by Merck & Co.), Vfend (voriconazole, marketed by Pfizer, Inc.), Sporanox (itraconazole, marketed by Jansen Pharmaceuticals, Inc.), Cresemba (isavuconazole, marketed by Astellas Pharma US, Inc.), Ambisome (liposomal amphothericin B, marketed by Astellas Pharma US, Inc.) Abelcet (lipid complex amphothericin B, marketed by Sigma Tau Phrmaceuticals Inc.) and amphothericin B deoxycholate (marketed by X-Gen Pharmaceuticals, Inc.). There currently are and may be more generic versions of these products available at the time of MAT2203 market approval, which will create added competition. In addition to approved therapies, we expect that MAT2203 may compete with product candidates that we are aware of in clinical development by third parties, such as SCY-078 (being developed by Scynexis, Inc.), CD101 (being developed by Cidara Therapeutics, Inc.) and certain products being developed by Viamet Pharmaceuticals Holdings, LLC, Vical Incorporated and F2G, Ltd.
| S-20 |
Manufacturing
We currently contract with one third party manufacturer to supply us with certain of the intermediates used in MAT9001 and a second manufacturer to formulate a third intermediate and supply us with the final drug form. We have a third manufacturer which fills and provides our final MAT9001 capsules. If any of these manufacturers should become unavailable to us for any reason, we have identified a number of potential replacements, although we might incur some delay in qualifying such replacements. We expect to add additional suppliers and manufacturers for both the intermediates and final MAT9001 drug product as we advance MAT9001 further into clinical development.
We currently lease and operate in-house manufacturing capabilities for our lead LNC platform delivery technology product candidate, MAT2203, and for our LNC platform discovery programs in the gene therapy and vaccine spaces. While sufficient to produce the clinical supplies of product necessary to conduct our ongoing clinical trials and potentially early commercialization of MAT2203, we may need to expand our internal manufacturing capabilities in the future. If we are not able to retain our current manufacturing facilities and if we do not develop additional in-house manufacturing capability for our MAT2203 and product candidates sufficient to produce product for commercialization of these products, we will need to develop relationships with third-party manufacturers for the manufacture of our product candidates which could be time consuming and expensive.
There are a number of potential third-party suppliers for amphotericin B, the generic active pharmaceutical ingredients in our lead clinical stage product candidate – MAT2203. Although to date we have not entered into formal supply agreements to secure sufficient supply of amphotericin B to support our clinical programs for MAT2203, we believe we will be able to secure supply of amphotericin B to support our clinical programs for MAT2203 and from one or more third-party suppliers. As we move through development for our product candidate, we expect to enter into long term supply arrangements for key active pharmaceutical ingredients.
Sales and Marketing
We currently do not have any sales and marketing infrastructure. We plan to retain U.S. marketing and sales rights or co-promotion rights for our product candidates for which we receive marketing approvals, particularly in situations where it is possible to access the market through a focused, specialized sales force. For situations in which a large sales force is required to access the market, and with respect to markets outside the United States, we generally plan to commercialize our product candidates through collaborative arrangements with leading pharmaceutical and biotechnology companies.
Implications of Being an Emerging Growth Company
We are an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act of 2012. We will remain an emerging growth company until December 31, 2019, or until the earliest of (i) the last day of the first fiscal year in which our annual gross revenues exceed $1.07 billion, (ii) the date that we become a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act, which would occur if the market value of our common stock that is held by non-affiliates exceeds $700 million as of the last business day of our most recently completed second fiscal quarter, or (iii) the date on which we have issued more than $1 billion in non-convertible debt during the preceding three year period.
| S-21 |
For as long as we remain an “emerging growth company,” we intend to take advantage of certain exemptions from various reporting requirements that are applicable to public companies that are not “emerging growth companies” including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation and financial statements in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote to approve executive compensation and shareholder approval of any golden parachute payments not previously approved. We will take advantage of these reporting exemptions until we are no longer an “emerging growth company.”
Corporate Information
We were incorporated in Delaware under the name Matinas BioPharma Holdings, Inc. in May 2013. We have two operating subsidiaries: Matinas BioPharma, Inc., a Delaware corporation, and Matinas BioPharma Nanotechnologies, Inc., a Delaware corporation. Nereus BioPharma LLC, a Delaware limited liability company (and Matinas BioPharma’s predecessor) was formed on August 12, 2011. On February 29, 2012, Nereus BioPharma LLC converted from a limited liability company to a corporation and changed its name to Matinas BioPharma, Inc. In July 2013, Matinas BioPharma, Inc. merged with and into a wholly-owned subsidiary of ours, thereby becoming a wholly owned subsidiary of ours. On January 29, 2015, we acquired Aquarius Biotechnologies Inc. which was subsequently renamed Matinas BioPharma Nanotechnologies, Inc.
Our principal executive offices are located at 1545 Route 206 South, Suite 302, Bedminster, New Jersey 07921, and our telephone number is (908) 443-1860. Our website address is www.matinasbiopharma.com. Our website and the information contained on, or that can be accessed through, our website will not be deemed to be incorporated by reference in, and are not considered part of, this prospectus. You should not rely on our website or any such information in making your decision whether to purchase our securities.
Recent Results
At December 31, 2018, the Company had approximately $13.0 million in cash and cash equivalents, including restricted cash. This amount is preliminary, unaudited and subject to the completion of the audit of the Company’s consolidated financial statements as of and for the year ended December 31, 2018 (Audited 2018 Financial Statements). As a result, this amount may differ from the amount that will be reflected in the Audited 2018 Financial Statements. Additional information and disclosures are required for a more complete understanding of the Company’s financial position and results of operations as of December 31, 2018.
| S-22 |
| Common stock offered by us | 27,272,727 shares of common stock | |
| Common stock to be outstanding immediately after the offering | 124,969,970 shares | |
| Use of proceeds | We intend to use the net proceeds from this offering primarily for ongoing development activities for our product candidates, namely MAT9001, and our LNC platform delivery technology and for working capital and other general corporate purposes. See “Use of Proceeds.” | |
| Risk factors | Investing in our securities involves a high degree of risk. See “Risk Factors” beginning on page S-24 of this prospectus supplement and under similar headings in the other documents that are filed after the date hereof and incorporated by reference in this prospectus supplement for a discussion of factors to consider before deciding to purchase any of our securities. | |
| NYSE American symbol | “MTNB”. |
The number of shares of common stock to be outstanding after this offering is based on 97,697,243 shares of common stock outstanding on September 30, 2018 and excludes:
| ● | 11,455,029 shares issuable upon the exercise of outstanding stock options with a weighted-average exercise price of $1.21 per share; | |
| ● | 14,678,580 shares issuable upon conversion of outstanding shares of Series A Preferred Stock; | |
| ● | 2,348,573 shares issuable upon payment of dividends on Series A Preferred Stock; | |
| ● | 14,006,000 shares issuable upon conversion of outstanding shares of Series B Preferred Stock; | |
| ● | 5,802,256 shares issuable upon the exercise of outstanding warrants with a weighted-average exercise price of $0.61 per share; | |
| ● | 4,890,183 shares available for future issuance under our equity compensation plans; and | |
| ● | 3,000,000 shares issuable upon achievement of certain milestones pursuant to the terms of our merger agreement with Aquarius Biotechnologies, Inc. |
Except as otherwise noted, all information in this prospectus assumes no exercise by the underwriters of their option to purchase additional shares of common stock from us.
| S-23 |
An investment in our common stock involves a high degree of risk. You should carefully consider the risks described under “Risk Factors” in the accompanying prospectus and our Annual Report on Form 10-K for the year ended December 31, 2017 and any subsequent updates described in our Quarterly Reports on Form 10-Q and Current Reports on Form 8-K, all of which are incorporated herein by reference, and as updated by any other document that we subsequently file with the Securities and Exchange Commission and that is incorporated by reference into this prospectus supplement and the accompanying prospectus, as well as the risks described below and all of the other information contained in this prospectus supplement and the accompanying prospectus, and incorporated by reference into this prospectus supplement and the accompanying prospectus, including our financial statements and related notes, before investing in our securities. These risks and uncertainties are not the only ones facing us and there may be additional matters that we are unaware of or that we currently consider immaterial. All of these could adversely affect our business, business prospects, cash flow, results of operations and financial condition. In such case, the trading price of our common stock could decline, and you could lose all or part of your investment in our common stock.
Risks Related to Our Financial Position and Need for Additional Capital
We have incurred significant losses since our inception. We expect to incur losses over the next several years and may never achieve or maintain profitability.
We have incurred significant operating losses in every year since inception and expect to incur net operating losses for the foreseeable future. Our net loss was $15.5 million, $7.6 million and $9.1 million for the years ended December 31, 2017, 2016 and 2015, respectively. As of September 30, 2018, we had an accumulated deficit of $62.3 million. We do not know whether or when we will become profitable. To date, we have not generated any revenues from product sales and have financed our operations primarily through private placements of our equity securities and, to a lesser extent, through funding from the National Institutes of Health, or the NIH. We have devoted substantially all of our financial resources and efforts to the research and development of potential product candidates. We are still in the early stages of development of our product candidates, and we have not completed development of any product candidate. We expect to continue to incur significant expenses and operating losses over the next several years. Our net losses may fluctuate significantly from quarter to quarter and year to year. Net losses and negative cash flows have had, and will continue to have, an adverse effect on our stockholders’ deficit and working capital. We anticipate that our expenses will increase substantially if and as we:
| ● | conduct further preclinical and clinical studies of MAT9001, our lead product candidate; | |
| ● | support the conduct of further clinical studies of MAT2203, even if such studies are primarily financed with non-dilutive funding from the NIH; | |
| ● | seek to discover and develop additional product candidates; | |
| ● | seek regulatory approvals for any product candidates that successfully complete clinical trials; | |
| ● | require the manufacture of larger quantities of product candidates for clinical development and potentially commercialization; | |
| ● | maintain, expand and protect our intellectual property portfolio; | |
| ● | hire additional clinical, quality control and scientific personnel; and | |
| ● | add operational, financial and management information systems and personnel, including personnel to support our product development and planned future commercialization efforts and personnel and infrastructure necessary to help us comply with our obligations as a public company. |
Our ability to become and remain profitable depends on our ability to generate revenue. We do not expect to generate significant revenue unless and until we are able to obtain marketing approval for, and successfully commercialize, one or more of our product candidates. This will require us to be successful in a range of challenging activities, including completing preclinical testing and clinical trials of our product candidates, discovering additional product candidates, obtaining regulatory approval for these product candidates, manufacturing, marketing and selling any products for which we may obtain regulatory approval, satisfying any post-marketing requirements and obtaining reimbursement for our products from private insurance or government payors. We are only in the preliminary stages of most of these activities and have not yet commenced other of these activities. We may never succeed in these activities and, even if we do, may never generate revenues that are significant enough to achieve profitability.
| S-24 |
Because of the numerous risks and uncertainties associated with pharmaceutical product development, we are unable to accurately predict the timing or amount of increased expenses or when, or if, we will be able to achieve profitability. If we are required by the U.S. Food and Drug Administration, or the FDA, or comparable non-U.S. regulatory authorities to perform studies in addition to those currently expected, or if there are any delays in completing our clinical trials or the development of any of our product candidates, our expenses could increase.
Even if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. Our failure to become and remain profitable would decrease the value of our company and could impair our ability to raise capital, expand our business, maintain our research and development efforts, diversify our pipeline of product candidates or even continue our operations. A decline in the value of our company could also cause you to lose all or part of your investment.
Our recurring losses from operations have raised substantial doubt regarding our ability to continue as a going concern.
Our recurring losses from operations raise substantial doubt about our ability to continue as a going concern, and as a result, our independent registered public accounting firm included an explanatory paragraph in its report on our financial statements as of and for the year ended December 31, 2017 with respect to this uncertainty. This going concern opinion, and any future going concern opinion, could materially limit our ability to raise additional capital. We have incurred significant losses since our inception and have never been profitable, and it is possible we will never achieve profitability. To date, we have devoted our resources to developing MAT9001 and our lead anti-infective product candidates, MAT2203 and MAT2501 and other product candidates developed from our cochleate drug delivery platform technology, but none of these product candidates can be marketed until regulatory approval has been obtained. Meaningful revenues will likely not be available until, and unless, MAT9001, MAT2203 or any of our other product candidates are approved by the FDA or comparable regulatory agencies in other countries and successfully marketed, either by us, or a partner. The perception that we may not be able to continue as a going concern may cause potential partners or investors to choose not to deal with us due to concerns about our ability to meet our contractual and financial obligations.
We will need substantial additional funding. If we are unable to raise capital when needed, we could be forced to delay, reduce or eliminate our product development programs or commercialization efforts.
We expect our expenses to increase in connection with our ongoing activities, particularly as we conduct additional preclinical and clinical studies of MAT9001, our lead product candidate, as well as the anticipated Phase 2 clinical trial of MAT2203 in cryptococcal meningitis, conduct additional preclinical and clinical trials to further validate and expand our LNC platform delivery technology, continue research and development, initiate clinical trials and, if development succeeds, seek regulatory approval of our product candidates. Our expenses could further increase if we initiate new research and preclinical development efforts for other product candidates. In addition, if we obtain regulatory approval for any of our product candidates, we expect to incur significant commercialization expenses related to product manufacturing, marketing, sales and distribution. Furthermore, we expect to incur significant additional costs associated with operating as a public company, particularly as we cease to qualify as an “emerging growth company.” Accordingly, we will need to obtain substantial additional funding in connection with our continuing operations. If we are unable to raise capital when needed or on attractive terms, we could be forced to delay, reduce or eliminate our research and development programs or any future commercialization efforts.
We believe that our existing cash and cash equivalents, including restricted cash, of approximately $13.0 million as of December 31, 2018, will enable us to fund our operating expenses and capital expenditure requirements into January of 2020. We have based this estimate on assumptions that may prove to be wrong in the future, and we could use our capital resources sooner than we currently expect. Changing circumstances could cause us to consume capital significantly faster than we currently anticipate, and we may need to spend more money than currently expected because of circumstances beyond our control. Our future capital requirements, both short-term and long-term, will depend on many factors, including:
| S-25 |
| ● | the progress, timing, costs and results of our ongoing and planned clinical trials of MAT9001; | |
| ● | the scope, progress, timing, costs and results of clinical trials of, and research and preclinical development efforts for, other product candidates, including MAT2203, any future product candidates based upon our cochleate delivery technology platform, and any preclinical or clinical work done to further validate our cochleate platform delivery technology, generally; | |
| ● | our ability to enter into and the terms and timing of any collaborations, licensing or other arrangements that we may establish; | |
| ● | the number and development requirements of other product candidates that we pursue; | |
| ● | the costs, timing and outcome of regulatory review of our product candidates by the FDA and comparable non-U.S. regulatory authorities; | |
| ● | the costs and timing of future commercialization activities, including product manufacturing, marketing, sales and distribution, for any of our product candidates for which we receive marketing approval; | |
| ● | the revenue, if any, received from commercial sales of our product candidates for which we receive marketing approval; | |
| ● | our headcount growth and associated costs as we expand our research and development and establish a commercial infrastructure; | |
| ● | the costs and timing of preparing, filing and prosecuting patent applications, maintaining and enforcing our intellectual property rights and defending any intellectual property-related claims; | |
| ● | the extent to which we acquire or in-license other products and technologies; | |
| ● | the costs of operating as a public company; and | |
| ● | the effect of competing technological and market developments. |
Identifying potential product candidates and conducting preclinical testing and clinical trials is a time-consuming, expensive and uncertain process that takes years to complete, and we may never generate the necessary data or results required to obtain regulatory approval and achieve product sales. In addition, our product candidates, if approved, may not achieve commercial success. Our commercial revenues, if any, will be derived from sales of products that we do not expect to be commercially available for many years, if at all. Accordingly, we will need to continue to rely on additional financing to achieve our business objectives. Adequate additional financing may not be available to us on acceptable terms, or at all. In addition, we may seek additional capital due to favorable market conditions or strategic considerations, even if we believe we have sufficient funds for our current or future operating plans.
Raising additional capital may cause dilution to our stockholders, restrict our operations or require us to relinquish rights to our technologies or product candidates.
Until such time, if ever, as we can generate product revenues sufficient to achieve profitability, we expect to finance our cash needs through a combination of public or private equity offerings, debt financings, government or other third party funding, collaborations and licensing arrangements. We do not have any committed external source of funds other than limited grant funding from the NIH. To the extent that we raise additional capital through the sale of common stock, convertible securities or other equity securities, your ownership interest may be materially diluted, and the terms of these securities may include liquidation or other preferences and anti-dilution protections that could adversely affect your rights as a common stockholder. Debt financing and preferred equity financing, if available, would result in increased fixed payment obligations and may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends, that could adversely impact our ability to conduct our business. Securing additional financing could require a substantial amount of time and attention from our management and may divert a disproportionate amount of their attention away from day-to-day activities, which may adversely affect our management’s ability to oversee the development of our product candidates.
If we raise additional funds through collaborations, strategic alliances or marketing, distribution or licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies, future revenue streams, research programs or product candidates or grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to delay, limit, reduce or terminate our product development or future commercialization efforts or grant rights to develop and market product candidates that we would otherwise prefer to develop and market ourselves.
| S-26 |
Our stockholders may be subject to substantial dilution by exercises of outstanding options and warrants, conversion of preferred shares and by the future issuance of common stock to the former stockholders of Aquarius pursuant to the terms of the merger agreement.
As of September 30, 2018, we had outstanding options to purchase an aggregate of 11,455,029 shares of our common stock at a weighted average exercise price of $1.21 per share and warrants to purchase an aggregate of 5,802,256 shares of our common stock at a weighted average exercise price of $0.61 per share. In addition, as of September 30, 2018, we had 1,467,858 million shares of Series A Preferred Stock outstanding and 7,003 shares of Series B Preferred Stock outstanding. Each share of Series A Preferred Stock may be converted into 10 shares of common stock upon the request of the holder and each share of Series B Preferred Stock may be converted into 2,000 shares of common stock upon the request of the holder. The conversion of preferred shares and the exercise of such outstanding options and the warrants, will result in dilution of the value of our shares. In addition, pursuant to the terms of the merger agreement with Matinas BioPharma Nanotechnologies, Inc. (f/k/a Aquarius Biotechnologies, Inc.), we will be required to issue up to an additional 3,000,000 shares of our common stock upon the achievement of certain milestones. The milestone consideration consists of (i) 1,500,000 shares issuable upon the dosing of the first patient in a phase III trial sponsored by us for a product utilizing the cochleate delivery technology and (ii) 1,500,000 shares issuable upon FDA approval of the first NDA submitted by us for a product utilizing the cochleate delivery technology.
Our limited operating history may make it difficult for you to evaluate the success of our business to date and to assess our future viability.
We commenced active operations in 2013 and have a limited operating history. Our product candidates are in early stages of clinical development. We have not yet demonstrated our ability to successfully obtain regulatory approvals for any of our product candidates, manufacture a commercial scale product, or arrange for a third party to do so on our behalf, or conduct sales and marketing activities necessary for successful product commercialization. Consequently, any predictions you make about our future success or viability may not be as accurate as they could be if we had a longer operating history.
In addition, as a young business, we may encounter unforeseen expenses, difficulties, complications, delays and other known and unknown factors. Even if we obtain regulatory approval, we will need to transition from a company with a research and development focus to a company capable of supporting commercial activities. We may not be successful in such a transition.
We expect our financial condition and operating results to continue to fluctuate significantly from quarter-to-quarter and year-to-year due to a variety of factors, many of which are beyond our control. Accordingly, you should not rely upon the results of any quarterly or annual periods as indications of future operating performance.
U.S. federal income tax reform could materially affect our tax obligations and effective tax rate.
On December 22, 2017, the Tax Cuts and Jobs Act, or the Tax Act, was signed into law, significantly reforming the tax code. The Tax Act, among other things, includes changes to U.S. federal tax rates, imposes significant additional limitations on the deductibility of interest, limits net operating loss (NOL) deductions, allows for the expensing of capital expenditures, puts into effect the migration from a “worldwide” system of taxation to a territorial system and modifies or repeals many business deductions and credits. The estimated impact of the Tax Act is based on our management’s current knowledge and assumptions, and recognized impacts could be materially different from current estimates based on our actual results and our further analysis of the new law. We have revalued our net deferred tax assets and liabilities at the newly enacted U.S. federal rate, and we recognized a tax benefit of $.4 million during the year ended December 31, 2017 related to the TCJA.
We continue to examine the impact this tax reform legislation may have on our business. The Tax Act requires complex computations not previously provided in U.S. tax law. As such, the application of accounting guidance for such items is currently uncertain. Further, compliance with the Tax Act and the accounting for such provisions require accumulation of information not previously required or regularly produced. As additional regulatory guidance is issued by the applicable taxing authorities, as accounting treatment is clarified, as we perform additional analysis on the application of the law, and as we refine estimates in calculating the effect, our final analysis, which will be recorded in the period completed, may be different from our current provisional amounts, which could materially affect our tax obligations and effective tax rate.
| S-27 |
Risks Related to Product Development, Regulatory Approval, Manufacturing and Commercialization
We are early in our development efforts, which may not be successful.
We completed a PK/PD study of MAT9001 head to head vs. Vascepa in 2015. We recently completed two separate Phase 2 clinical trials of MAT2203. Because of the early stage of our development efforts, we are still in the process of determining the overall clinical development path for our current and future product candidates. As a result, the timing and costs of the regulatory paths we will follow and marketing approvals remain uncertain. Our ability to generate product revenue, which we do not expect will occur for many years, if ever, will depend heavily on the successful development and eventual commercialization of our early-stage product candidates. The success of MAT9001, MAT2203, and any other product candidates we may develop will depend on many factors, including the following:
| ● | successful completion of preclinical studies; | |
| ● | successful enrollment in, and completion of, clinical trials; | |
| ● | demonstrating safety and efficacy; | |
| ● | receipt of marketing approvals from applicable regulatory authorities; | |
| ● | establishing clinical and commercial manufacturing capabilities or making arrangements with third-party manufacturers; | |
| ● | obtaining and maintaining patent and trade secret protection and non-patent exclusivity for our product candidates and technologies; | |
| ● | launching commercial sales of the product candidates, if and when approved, whether alone or selectively in collaboration with others; | |
| ● | acceptance of the product candidates, if and when approved, by patients, the medical community and third-party payers; | |
| ● | effectively competing with other therapies; | |
| ● | a continued acceptable safety profile of the products following approval; and | |
| ● | enforcing and defending intellectual property rights and claims. |
If we do not accomplish one or more of these goals in a timely manner, or at all, we could experience significant delays or an inability to successfully commercialize our product candidates, which would harm our business.
We cannot be certain that MAT9001, MAT2203 or any other product candidates that we may develop will receive regulatory approval, and without regulatory approval we will not be able to market any of our product candidates. Any delay in the regulatory review or approval of any of our product candidates will materially or adversely harm our business.
We expect to invest most of our capital in the development of MAT9001. Our ability to generate revenue related to product sales, which we do not expect will occur for at least the next several years, if ever, will depend on the successful development and regulatory approval of one or more of our product candidates. All of our product candidates require regulatory review and approval prior to commercialization. Any delays in the regulatory review or approval of our product candidates would delay market launch, increase our cash requirements and result in additional operating losses. This failure to obtain regulatory approvals would prevent our product candidate from being marketed and would have a material and adverse effect on our business.
| S-28 |
The process of obtaining FDA and other required regulatory approvals, including foreign approvals, often takes many years and can vary substantially based upon the type, complexity and novelty of the products involved. Furthermore, this approval process is extremely complex, expensive and uncertain. We may be unable to submit any new drug application, or an NDA, in the United States or any marketing approval application in foreign jurisdictions for any of our products. If we submit an NDA including any amended NDA or supplemental NDA, to the FDA seeking marketing approval for any of our product candidates, the FDA must decide whether to accept or reject the submission for filing. We cannot be certain that any of these submissions will be accepted for filing and reviewed by the FDA, or that the marketing approval application submissions to any other regulatory authorities will be accepted for filing and review by those authorities. We cannot be certain that we will be able to respond to any regulatory requests during the review period in a timely manner, or at all, without delaying potential regulatory action. We also cannot be certain that any of our product candidates will receive favorable recommendations from any FDA advisory committee or foreign regulatory bodies or be approved for marketing by the FDA or foreign regulatory authorities. In addition, delays in approvals or rejections of marketing applications may be based upon many factors, including regulatory requests for additional analyses, reports, data and studies, regulatory questions regarding data and results, changes in regulatory policy during the period of product development and the emergence of new information regarding such product candidates.
Data obtained from preclinical studies and clinical trials are subject to different interpretations, which could delay, limit or prevent regulatory review or approval of any of our product candidates. Furthermore, regulatory attitudes towards the data and results required to demonstrate safety and efficacy can change over time and can be affected by many factors, such as the emergence of new information, including on other products, policy changes and agency funding, staffing and leadership. We do not know whether future changes to the regulatory environment will be favorable or unfavorable to our business prospects.
In addition, the environment in which our regulatory submissions may be reviewed changes over time. For example, average review times at the FDA for NDAs have fluctuated over the last ten years, and we cannot predict the review time for any of our submissions with any regulatory authorities. Review times can be affected by a variety of factors, including budget and funding levels and statutory, regulatory and policy changes. Moreover, in light of widely publicized events concerning the safety risk of certain drug products, regulatory authorities, members of the U.S. Government Accountability Office, medical professionals and the general public have raised concerns about potential drug safety issues. These events have resulted in the withdrawal of drug products, revisions to drug labeling that further limit use of the drug products and establishment of REMS measures that may, for instance, restrict distribution of drug products. The increased attention to drug safety issues may result in a more cautious approach by the FDA to clinical trials. Data from clinical trials may receive greater scrutiny with respect to safety, which may make the FDA or other regulatory authorities more likely to terminate clinical trials before completion, or require longer or additional clinical trials that may result in substantial additional expense and a delay or failure in obtaining approval or may result in approval for a more limited indication than originally sought.
We depend in part on technology owned or licensed to us by third parties, and the loss of access to this technology would terminate or delay the further development of our product candidates, injure our reputation or force us to pay higher royalties.
We rely partially on the LNC platform delivery technology that we have licensed from Rutgers. The loss of access to this technology could materially impair our business and future viability, and could result in delays in developing, introducing or maintaining our product candidates and formulations until equivalent technology, if available, is identified, licensed and integrated. In addition, any defects in the technology we license could prevent the implementation or impair the functionality of our product candidates or formulation, delay new product or formulation introductions or injure our reputation. If we are required to enter into license agreements with third parties for replacement technology, we could be subject to higher royalty payments.
Clinical drug development involves a lengthy and expensive process with uncertain outcomes that may lead to delayed timelines and increased cost, and may prevent us from being able to complete clinical trials.
Clinical testing is expensive, can take many years to complete, and its outcome is inherently uncertain. The results of preclinical and clinical studies of our product candidates may not be predictive of the results of later-stage clinical trials. For example, the positive results generated to date in Phase 2 clinical studies for MAT9001 do not ensure that our Phase 3 clinical trials will demonstrate similar results. Product candidates in later stages of clinical trials may fail to show the desired safety and efficacy despite having progressed through preclinical studies and initial clinical trials. A number of companies in the pharmaceutical industry have suffered significant setbacks in advanced clinical trials due to lack of efficacy or adverse safety profiles, notwithstanding promising results in earlier studies, and we cannot be certain that we will not face similar setbacks.
| S-29 |
We cannot be certain that future clinical trials for MAT9001, MAT2203 or any of our other product candidates, will begin on time, not need to be redesigned, enroll an adequate number of patients on time or be completed on schedule, if at all, or that any interim analyses with respect to such trials will be completed on schedule or support continued clinical development of the associated product candidate.
We could also encounter delays if a clinical trial is suspended or terminated by us upon recommendation of the data monitoring committee for such trial, by the IRBs of the institutions in which such trials are being conducted, or by the FDA or other regulatory authorities. Such authorities may suspend or terminate a clinical trial due to a number of factors, including failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols, inspection of the clinical trial operations or trial site by the FDA or other regulatory authorities resulting in the imposition of a clinical hold, unforeseen safety issues or adverse side effects, failure to demonstrate a benefit from using a drug, changes in governmental regulations or administrative actions, or lack of adequate funding to continue the clinical trial.
If we experience delays in the completion of, or termination of, any clinical trial of our product candidates, the commercial prospects of our product candidates may be harmed, and our ability to generate revenue from the sale of any of these product candidates will be delayed. In addition, any delays in completing our clinical trials will increase our costs, slow down our product candidate development and approval processes, and jeopardize our ability to commence product sales and generate revenue. Any of these occurrences may significantly harm our business, financial condition and prospects significantly.
Delays in the commencement, enrollment and completion of our clinical trials could result in increased costs to us and delay or limit our ability to obtain regulatory approval for MAT9001 and our other product candidates.
Delays in the commencement, enrollment and completion of clinical trials could increase our product development costs or limit the regulatory approval of our product candidates. The commencement, enrollment and completion of clinical trials can be delayed for a variety of reasons, including:
| ● | inability to reach agreements on acceptable terms with prospective contract research organizations (CROs) and trial sites, the terms of which can be subject to extensive negotiation and may vary significantly among different CROs and trial sites; | |
| ● | inability to maintain necessary supplies of study drug and comparator to maintain predicted enrollment rates at clinical trial sites; | |
| ● | regulatory objections to commencing a clinical trial; | |
| ● | inability to identify and maintain a sufficient number of trial sites, many of which may already be engaged in other clinical trial programs, including some that may be for the same indication as our product candidates; | |
| ● | withdrawal of clinical trial sites from our clinical trials as a result of changing standards of care or the ineligibility of a site to participate in our clinical trials; | |
| ● | inability to obtain institutional review board approval, including that within the NIH, to conduct a clinical trial; | |
| ● | difficulty recruiting and enrolling subjects to participate in clinical trials for a variety of reasons, including meeting the enrollment criteria for our study and competition from other clinical trial programs for the same indication as our product candidates; | |
| ● | inability to retain subjects in clinical trials due to the treatment protocol, personal issues, side effects from the therapy or lack of efficacy; and | |
| ● | difficulty in importing and exporting clinical trial materials and study samples. |
| S-30 |
We may not have or be able to obtain sufficient quantities of our products to meet our supply and clinical studies obligations and our business, financial condition and results of operation may be adversely affected.
To date, we have only developed limited in-house manufacturing capabilities for the LNC technology needed for the clinical development our MAT2203 product candidate and rely exclusively on third party manufacturers for the manufacture of MAT9001. If we do not develop a long term in-house manufacturing capability for the cochleates needed for our product candidates sufficient to produce product for continued development and, if regulatory approval is obtained, then commercialization of these products, we will be dependent on a small number of third-party manufacturers for the manufacture of our product candidates. We may not have long-term agreements with any of these third parties, and if they are unable or unwilling to perform for any reason, we may not be able to locate alternative acceptable manufacturers or formulators or enter into favorable agreements with them. Any inability to acquire sufficient quantities of our products in a timely manner from these third parties could delay clinical trials and prevent us from developing our products in a cost-effective manner or on a timely basis. In addition, manufacturers of our product candidates are subject to cGMP and similar foreign standards and we would not have control over compliance with these regulations by our manufacturers. If one of our contract manufacturers fails to maintain compliance, the production of our products could be interrupted, resulting in delays and additional costs. In addition, if the facilities of such manufacturers do not pass a pre-approval or post-approval plant inspection, the FDA will not grant approval and may institute restrictions on the marketing or sale of our products.
We may be reliant on third party manufactures and suppliers to meet the demands of our clinical supplies. Delays in receipt of materials, scheduling, release, custom’s control, and regulatory compliance issues may adversely impact our ability to initiate, maintain, or complete clinical trials that we are sponsoring. Commercial manufacturing and supply agreements have not been established. Issues arising from scale-up, environmental controls, equipment requirements, or other factors, may have an adverse impact on our ability to manufacture our product candidates.
Even if we obtain regulatory approval for our product candidates, if we are unable to successfully commercialize our products, it will limit our ability to generate revenue and will materially adversely affect our business, financial condition and results of operations.
Even if we obtain regulatory approval for our product candidates, our long-term viability and growth depend on the successful commercialization of products which lead to revenue and profits. Pharmaceutical product development is an expensive, high risk, lengthy, complicated, resource intensive process. In order to succeed, among other things, we must be able to:
| ● | identify potential drug product candidates; | |
| ● | design and conduct appropriate laboratory, preclinical and other research; | |
| ● | submit for and receive regulatory approval to perform clinical studies; | |
| ● | design and conduct appropriate preclinical and clinical studies according to good laboratory and good clinical practices; | |
| ● | select and recruit clinical investigators; | |
| ● | select and recruit subjects for our studies; | |
| ● | collect, analyze and correctly interpret the data from our studies; | |
| ● | submit for and receive regulatory approvals for marketing; and | |
| ● | manufacture the drug product candidates according to cGMP. |
The development program with respect to any given product will take many years and thus delay our ability to generate profits. In addition, potential products that appear promising at early stages of development may fail for a number of reasons, including the possibility that the products may require significant additional testing or turn out to be unsafe, ineffective, too difficult or expensive to develop or manufacture, too difficult to administer, or unstable. Failure to successfully commercialize our products will adversely affect our business, financial condition and results of operations.
| S-31 |
If our preclinical and clinical studies do not produce positive results, if our clinical trials are delayed or if serious side effects are identified during such studies or trials, we may experience delays, incur additional costs and ultimately be unable to commercialize our product candidates.
Before obtaining regulatory approval for the sale of our product candidates, we must conduct, generally at our own expense, extensive preclinical tests to demonstrate the safety of our product candidates in animals, and clinical trials to demonstrate the safety and efficacy of our product candidates in humans. Preclinical and clinical testing is expensive, difficult to design and implement and can take many years to complete. A failure of one or more of our preclinical studies or clinical trials can occur at any stage of testing. We may experience numerous unforeseen events during, or as a result of, preclinical testing and the clinical trial process that could delay or prevent our ability to obtain regulatory approval or commercialize our product candidates, including:
| ● | our preclinical tests or clinical trials may produce negative or inconclusive results, and we may decide, or regulators may require us, to conduct additional preclinical testing or clinical trials or we may abandon projects that we expect to be promising; | |
| ● | regulators or institutional review boards may not authorize us to commence a clinical trial or conduct a clinical trial at a prospective trial site; | |
| ● | conditions imposed on us by the FDA or any non-U.S. regulatory authority regarding the scope or design of our clinical trials may require us to resubmit our clinical trial protocols to institutional review boards for re-inspection due to changes in the regulatory environment; | |
| ● | the number of patients required for our clinical trials may be larger than we anticipate or participants may drop out of our clinical trials at a higher rate than we anticipate; | |
| ● | our third-party contractors or clinical investigators may fail to comply with regulatory requirements or fail to meet their contractual obligations to us in a timely manner; | |
| ● | we might have to suspend or terminate one or more of our clinical trials if we, the regulators or the institutional review boards determine that the participants are being exposed to unacceptable health risks; | |
| ● | regulators or institutional review boards may require that we hold, suspend or terminate clinical research for various reasons, including noncompliance with regulatory requirements; | |
| ● | the cost of our clinical trials may be greater than we anticipate; | |
| ● | the supply or quality of our product candidates or other materials necessary to conduct our clinical trials may be insufficient or inadequate or we may not be able to reach agreements on acceptable terms with prospective clinical research organizations; and | |
| ● | the effects of our product candidates may not be the desired effects or may include undesirable side effects or the product candidates may have other unexpected characteristics. |
In addition, if we are required to conduct additional clinical trials or other testing of our product candidates beyond those that we currently contemplate, if we are unable to successfully complete our clinical trials or other testing, if the results of these trials or tests are not positive or are only modestly positive or if there are safety concerns, we may:
| ● | be delayed in obtaining, or may not be able to obtain, marketing approval for one or more of our product candidates; | |
| ● | obtain approval for indications that are not as broad as intended or entirely different than those indications for which we sought approval; or | |
| ● | have the product removed from the market after obtaining marketing approval. |
Our product development costs will also increase if we experience delays in testing or approvals. We do not know whether any preclinical tests or clinical trials will be initiated as planned, will need to be restructured or will be completed on schedule, if at all. Significant preclinical or clinical trial delays also could shorten the patent protection period during which we may have the exclusive right to commercialize our product candidates. Such delays could allow our competitors to bring products to market before we do and impair our ability to commercialize our products or product candidates.
| S-32 |
If we cannot enroll enough patients to complete our clinical trials, such failure may adversely affect our business, financial condition and results of operations.
The completion rate of clinical studies of our products is dependent on, among other factors, the patient enrollment rate. Patient enrollment is a function of many factors, including:
| ● | investigator identification and recruitment; | |
| ● | regulatory approvals to initiate study sites; | |
| ● | patient population size; | |
| ● | the nature of the protocol to be used in the trial; | |
| ● | patient proximity to clinical sites; | |
| ● | eligibility criteria for the study; | |
| ● | competition from other companies’ clinical studies for the same patient population; and | |
| ● | ability to obtain comparator drug/device. |
We believe our procedures for enrolling patients have been appropriate; however, delays in patient enrollment would increase costs and delay ultimate commercialization and sales, if any, of our products. Such delays could materially adversely affect our business, financial condition and results of operations.
If we are unsuccessful in identifying and developing additional product candidates, our potential for growth may be impaired.
Even if we receive regulatory approval for MAT9001, MAT2203 or any other product candidates we may develop, we still may not be able to successfully commercialize such products and the revenue that we generate from its sales, if any, may be limited.
If approved for marketing, the commercial success of MAT9001, MAT2203 or any other product candidates we may develop will depend upon its acceptance by the medical community, including physicians, patients and health care payors. The degree of market acceptance of MAT9001, MAT2203 or such other product candidate will depend on a number of factors, including:
| ● | demonstration of clinical safety and efficacy of such product candidate; | |
| ● | relative convenience and ease of administration; | |
| ● | the prevalence and severity of any adverse effects; | |
| ● | the willingness of physicians to prescribe such product candidates and of the target patient population to try new therapies; | |
| ● | pricing and cost-effectiveness; | |
| ● | the inclusion or omission of such product candidate in applicable treatment guidelines; | |
| ● | the effectiveness of our or any future collaborators’ sales and marketing strategies; | |
| ● | limitations or warnings contained in FDA-approved labeling; | |
| ● | our ability to obtain and maintain sufficient third-party coverage or reimbursement from government health care programs, including Medicare and Medicaid, private health insurers and other third-party payors; and | |
| ● | the willingness of patients to pay out-of-pocket in the absence of third-party coverage or reimbursement. |
If MAT9001, MAT2203, or any other product candidates we may develop is approved, but does not achieve an adequate level of acceptance by physicians, health care payors and patients, we may not generate sufficient revenue and we may not be able to achieve or sustain profitability. Our efforts to educate the medical community and third-party payors on the benefits of such product candidate may require significant resources and may never be successful.
| S-33 |
In addition, even if we obtain regulatory approvals, the timing or scope of any approvals may prohibit or reduce our ability to commercialize such product candidate successfully. For example, if the approval process takes too long, we may miss market opportunities and give other companies the ability to develop competing products or establish market dominance. Any regulatory approval we ultimately obtain may be limited or subject to restrictions or post-approval commitments that render such product candidate not commercially viable. For example, regulatory authorities may approve such product candidate for fewer or more limited indications than we request, may not approve the price we intend to charge for such product candidate, may grant approval contingent on the performance of costly post-marketing clinical trials, or may approve such product candidate with a label that does not include the labeling claims necessary or desirable for the successful commercialization of that indication. Further, the FDA may place conditions on approvals including potential requirements or risk management plans and the requirement for a Risk Evaluation and Mitigation Strategy (“REMS”) to assure the safe use of the drug. If the FDA concludes a REMS is needed, the sponsor of the NDA must submit a proposed REMS; the FDA will not approve the NDA without an approved REMS, if required. A REMS could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. Any of these limitations on approval or marketing could restrict the commercial promotion, distribution, prescription or dispensing of such product candidate. Moreover, product approvals may be withdrawn for non-compliance with regulatory standards or if problems occur following the initial marketing of the product. Any of the foregoing scenarios could materially harm the commercial success of such product candidate.
We currently have no sales and marketing organization. If we are unable to establish satisfactory sales and marketing capabilities, we may not successfully commercialize any of our product candidates, if regulatory approval is obtained.
At present, we have no sales or marketing personnel. In order to commercialize products that are approved for commercial sales, we must either develop a sales and marketing infrastructure or collaborate with third parties that have such commercial infrastructure. If we elect to develop our own sales and marketing organization, we do not intend to begin to hire sales and marketing personnel until the time of NDA submission to the FDA at the earliest, and we do not intend to establish our own sales organization in the United States until shortly prior to FDA approval of MAT9001, MAT2203 or any of our other product candidates.
We may not be able to establish a direct sales force in a cost-effective manner or realize a positive return on this investment. In addition, we will have to compete with established and well-funded pharmaceutical and biotechnology companies to recruit, hire, train and retain sales and marketing personnel. Factors that may inhibit our efforts to commercialize MAT9001, MAT2203 or any of our other product candidates in the United States without strategic partners or licensees include:
| ● | our inability to recruit and retain adequate numbers of effective sales and marketing personnel; | |
| ● | the inability of sales personnel to obtain access to or persuade adequate numbers of physicians to prescribe our future products; | |
| ● | the lack of complementary products to be offered by sales personnel, which may put us at a competitive disadvantage relative to companies with more extensive product lines; and | |
| ● | unforeseen costs and expenses associated with creating an independent sales and marketing organization. |
If we are not successful in recruiting sales and marketing personnel or in building a sales and marketing infrastructure, or if we do not successfully enter into appropriate collaboration arrangements, we will have difficulty successfully commercializing MAT9001, MAT2203 or any other product candidates we may develop, which would adversely affect our business, operating results and financial condition. Outside the United States, we may commercialize our product candidates by entering into collaboration agreements with pharmaceutical partners. We may not be able to enter into such agreements on terms acceptable to us or at all. In addition, even if we enter into such relationships, we may have limited or no control over the sales, marketing and distribution activities of these third parties. Our future revenues may depend heavily on the success of the efforts of these third parties
| S-34 |
If we are unable to file for approval of MAT9001 or MAT2203 under Section 505(b)(2) of the FDCA or if we are required to generate additional data related to safety and efficacy in order to obtain approval under Section 505(b)(2), we may be unable to meet our anticipated development and commercialization timelines.
Our current plans for filing the NDAs for MAT9001 and MAT2203 include efforts to minimize the data we will be required to generate in order to obtain marketing approval for this product candidate and therefore reduce the development time. Based upon written feedback received from the FDA in 2014, we believe this approach will create the opportunity for us to leverage existing data developed with certain existing omega-3 fatty acids to create a streamlined approach to potential approval for MAT9001 for the treatment of severe hypertriglyceridemia. Although our interactions with the FDA have encouraged our efforts to continue to develop MAT9001 for severe hypertriglyceridemia, there is no assurance that we will satisfy the FDA’s requirements for approval in this indication. Likewise, we intend to rely on the history of efficacy of amphotericin B, and anticipate meeting with the FDA in the first half of 2019 to discuss our development plans for MAT2203.The timelines for filing and review of our NDAs for MAT9001 and MAT2203 are based on our plan to submit these NDAs under Section 505(b)(2) of the FDCA, which would enable us to rely in part on data in the public domain or elsewhere. We have not yet filed an NDA under Section 505(b)(2) for any product candidate. Depending on the data that may be required by the FDA for approval, some of the data may be related to products already approved by the FDA. If the data relied upon is related to products already approved by the FDA and covered by third-party patents we would be required to certify that we do not infringe the listed patents or that such patents are invalid or unenforceable. As a result of the certification, the third-party would have 45 days from notification of our certification to initiate an action against us.
In the event that an action is brought in response to such a certification, the approval of our NDA could be subject to a stay of up to 30 months or more while we defend against such a suit. Approval of our product candidates under Section 505(b)(2) may therefore be delayed until patent exclusivity expires or until we successfully challenge the applicability of those patents to our product candidates. Alternatively, we may elect to generate sufficient additional clinical data so that we no longer rely on data which triggers a potential stay of the approval of our product candidates. Even if no exclusivity periods apply to our applications under Section 505(b)(2), the FDA has broad discretion to require us to generate additional data on the safety and efficacy of our product candidates to supplement third-party data on which we may be permitted to rely. In either event, we could be required, before obtaining marketing approval for any of our product candidates, to conduct substantial new research and development activities beyond those we currently plan to engage in order to obtain approval of our product candidates. Such additional new research and development activities would be costly and time consuming.
We may not be able to realize a shortened development timeline for MAT9001 for severe hypertriglyceridemia, and the FDA may not approve our NDA based on their review of the submitted data. If omega-3 fatty acids-containing products are withdrawn from the market by the FDA for any safety reason, we may not be able to reference such products to support a 505(b)(2) NDA for MAT9001and MAT2203, and we may need to fulfill the more extensive requirements of Section 505(b)(1). If we are required to generate additional data to support approval, we may be unable to meet our anticipated development and commercialization timelines, may be unable to generate the additional data at a reasonable cost, or at all, and may be unable to obtain marketing approval of our lead product candidate.
We face competition from other biotechnology and pharmaceutical companies and our operating results will suffer if we fail to compete effectively.
The biotechnology and pharmaceutical industries are intensely competitive and subject to rapid and significant technological change. We have competitors in a number of jurisdictions, many of which have substantially greater name recognition, commercial infrastructures and financial, technical and personnel resources than we have. We face competition from many different sources, including commercial pharmaceutical and biotechnology enterprises, academic institutions, government agencies and private and public research institutions. Established competitors may invest heavily to quickly discover and develop novel compounds that could make MAT9001, MAT2203 or any other product candidates we may develop obsolete or uneconomical. Any new product that competes with an approved product may need to demonstrate compelling advantages in efficacy, cost, convenience, tolerability and safety to be commercially successful. Other competitive factors, including generic competition, which could force us to lower prices or result in reduced sales, particularly those products that have been marketed by third parties for many years and are well accepted by physicians, patients and payers. In addition, new products developed by others could emerge as competitors to MAT9001, MAT2203 or any of our other product candidates. If we are not able to compete effectively against our current and future competitors, our business will not grow and our financial condition and operations will suffer.
| S-35 |
Further, although we believe that our proprietary LNC platform delivery technology, experience and knowledge in our areas of focus provide us with competitive advantages, potential competitors for MAT2203 could reduce our commercial opportunities.
Even if we obtain marketing approval for MAT9001, MAT2203 or any other product candidates that we may develop, we will be subject to ongoing obligations and continued regulatory review, which may result in significant additional expense. Additionally, our product candidates could be subject to labeling and other restrictions and withdrawal from the market and we may be subject to penalties if we fail to comply with regulatory requirements or if we experience unanticipated problems with our future products.
Even if we obtain United States regulatory approval of MAT9001, MAT2203 or any other product candidates that we may develop, FDA may still impose significant restrictions on its indicated uses or marketing or the conditions of approval, or impose ongoing requirements for potentially costly and time-consuming post-approval studies, and post-market surveillance to monitor safety and efficacy. Our future products will also be subject to ongoing regulatory requirements governing the manufacturing, labeling, packaging, storage, distribution, safety surveillance, advertising, promotion, recordkeeping and reporting of adverse events and other post-market information. These requirements include registration with FDA, as well as continued compliance with current Good Clinical Practices regulations, or cGCPs, for any clinical trials that we conduct post-approval. In addition, manufacturers of drug products and their facilities are subject to continuous review and periodic inspections by the FDA and other regulatory authorities for compliance with current good manufacturing practices, cGMP, requirements relating to quality control, quality assurance and corresponding maintenance of records and documents.
FDA has the authority to require a REMS, as part of an NDA or after approval, which may impose further requirements or restrictions on the distribution or use of an approved drug, such as limiting prescribing to certain physicians or medical centers that have undergone specialized training, limiting treatment to patients who meet certain safe-use criteria or requiring patient testing, monitoring and/or enrollment in a registry.
With respect to sales and marketing activities by us or any future partner, advertising and promotional materials must comply with FDA rules in addition to other applicable federal, state and local laws in the United States and similar legal requirements in other countries. In the United States, the distribution of product samples to physicians must comply with the requirements of the U.S. Prescription Drug Marketing Act. Application holders must obtain FDA approval for product and manufacturing changes, depending on the nature of the change. We may also be subject, directly or indirectly through our customers and partners, to various fraud and abuse laws, including, without limitation, the U.S. Anti-Kickback Statute, U.S. False Claims Act, and similar state laws, which impact, among other things, our proposed sales, marketing, and scientific/educational grant programs. If we participate in the U.S. Medicaid Drug Rebate Program, the Federal Supply Schedule of the U.S. Department of Veterans Affairs, or other government drug programs, we will be subject to complex laws and regulations regarding reporting and payment obligations. All of these activities are also potentially subject to U.S. federal and state consumer protection and unfair competition laws. Similar requirements exist in many of these areas in other countries.
In addition, our product labeling, advertising and promotion would be subject to regulatory requirements and continuing regulatory review. FDA strictly regulates the promotional claims that may be made about prescription products. In particular, a product may not be promoted for uses that are not approved by FDA as reflected in the product’s approved labeling. If we receive marketing approval for our product candidates, physicians may nevertheless legally prescribe our products to their patients in a manner that is inconsistent with the approved label. If we are found to have promoted such off-label uses, we may become subject to significant liability and government fines. FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant sanctions, including revocation of its marketing approval. The federal government has levied large civil and criminal fines against companies for alleged improper promotion and has enjoined several companies from engaging in off-label promotion. FDA has also requested that companies enter into consent decrees of permanent injunctions under which specified promotional conduct is changed or curtailed.
| S-36 |
If we or a regulatory agency discovers previously unknown problems with a product, such as adverse events of unanticipated severity or frequency, problems with the facility where the product is manufactured, or we or our manufacturers fail to comply with applicable regulatory requirements, we may be subject to the following administrative or judicial sanctions:
| ● | restrictions on the marketing or manufacturing of the product, withdrawal of the product from the market, or voluntary or mandatory product recalls; | |
| ● | issuance of warning letters or untitled letters; | |
| ● | clinical holds; | |
| ● | injunctions or the imposition of civil or criminal penalties or monetary fines; | |
| ● | suspension or withdrawal of regulatory approval; | |
| ● | suspension of any ongoing clinical trials; | |
| ● | refusal to approve pending applications or supplements to approved applications filed by us, or suspension or revocation of product license approvals; | |
| ● | suspension or imposition of restrictions on operations, including costly new manufacturing requirements; or | |
| ● | product seizure or detention or refusal to permit the import or export of product. |
The occurrence of any event or penalty described above may inhibit our ability to commercialize MAT9001, MAT2203 or any of our other product candidates and generate revenue. Adverse regulatory action, whether pre- or post-approval, can also potentially lead to product liability claims and increase our product liability exposure.
Future legislation, and/or regulations and policies adopted by the FDA may increase the time and cost required for us to conduct and complete clinical trials of MAT9001, MAT2203 and any other product candidates that we may develop.
FDA has established regulations to govern the drug development and approval process, as have foreign regulatory authorities. The policies of FDA and other regulatory authorities may change and additional laws or government regulations may be promulgated that could prevent, limit, delay but also accelerate regulatory review of our product candidates. For example, in December 2016, the Cures Act was signed into law. The Cures Act, among other things, is intended to modernize the regulation of drugs and spur innovation, but all of its provisions have yet to be implemented. Among other things, the Cures Act provides a new “limited population” pathway for certain antibacterial and antifungal drugs, or LPAD, but FDA has not issued final guidance regarding the LPAD yet. Additionally, in August 2017, FDA issued final guidance setting forth its current thinking with respect to development programs and clinical trial designs for antibacterial drugs to treat serous bacterial diseases in patients with an unmet medical need. We cannot predict what if any effect the Cures Act or any existing or future guidance from FDA will have on development of our product candidates.
Changes in health care law and implementing regulations, including government restrictions on pricing and reimbursement, as well as health care policy and other health care payor cost-containment initiatives, may have a material adverse effect on us.
In the United States and some foreign jurisdictions, there have been a number of legislative and regulatory changes and proposed changes regarding the healthcare system and efforts to control health care costs, including drug prices, that could have a significant negative impact on our business, including preventing, limiting or delay regulatory approval of our drug candidates and reducing the sales and profits derived from our products once they are approved.
For example, in the United States, the Patient Protection and Affordable Care Act of 2010 (“ACA”) substantially changed the way health care is financed by both governmental and private insurers and significantly affects the pharmaceutical industry. Many provisions of ACA impact the biopharmaceutical industry, including that in order for a biopharmaceutical product to receive federal reimbursement under the Medicare Part B and Medicaid programs or to be sold directly to U.S. government agencies, the manufacturer must extend discounts to entities eligible to participate in the drug pricing program under the Public Health Services Act, or PHS. Since its enactment, there have been judicial and Congressional challenges and amendments to certain aspects of ACA. There is continued uncertainty about the implementation of ACA, including the potential for further amendments to the ACA and legal challenges to or efforts to repeal the ACA.
| S-37 |
We cannot be sure whether additional legislative changes will be enacted, or whether government regulations, guidance or interpretations will be changed, or what the impact of such changes would be on the marketing approvals, sales, pricing, or reimbursement of our drug candidates or products, if any, may be.
Our future growth depends, in part, on our ability to penetrate foreign markets, where we would be subject to additional regulatory burdens and other risks and uncertainties.
Our future profitability will depend, in part, on our ability to commercialize our product candidates in foreign markets for which we intend to rely on collaborations with third parties. If we commercialize MAT9001, MAT2203 or any other product candidates that we may develop in foreign markets, we would be subject to additional risks and uncertainties, including:
| ● | our customers’ ability to obtain reimbursement for our product candidates in foreign markets; | |
| ● | our inability to directly control commercial activities because we are relying on third parties; | |
| ● | the burden of complying with complex and changing foreign regulatory, tax, accounting and legal requirements; | |
| ● | different medical practices and customs in foreign countries affecting acceptance in the marketplace; | |
| ● | import or export licensing requirements; | |
| ● | longer accounts receivable collection times; | |
| ● | longer lead times for shipping; | |
| ● | language barriers for technical training; | |
| ● | reduced protection of intellectual property rights in some foreign countries; | |
| ● | foreign currency exchange rate fluctuations; and | |
| ● | the interpretation of contractual provisions governed by foreign laws in the event of a contract dispute. |
Foreign sales of our product candidates could also be adversely affected by the imposition of governmental controls, political and economic instability, trade restrictions and changes in tariffs, any of which may adversely affect our results of operations.
If we market our product candidates in a manner that violates healthcare fraud and abuse laws, or if we violate government price reporting laws, we may be subject to civil or criminal penalties.
FDA enforces laws and regulations which require that the promotion of pharmaceutical products be consistent with the approved prescribing information. While physicians may prescribe an approved product for a so-called “off label” use, it is unlawful for a pharmaceutical company to promote its products in a manner that is inconsistent with its approved label and any company which engages in such conduct can subject that company to significant liability. Similarly, industry codes in the EU and other foreign jurisdictions prohibit companies from engaging in off-label promotion and regulatory agencies in various countries enforce violations of the code with civil penalties. While we intend to ensure that our promotional materials are consistent with our label, regulatory agencies may disagree with our assessment and may issue untitled letters, warning letters or may institute other civil or criminal enforcement proceedings. In addition to FDA restrictions on marketing of pharmaceutical products, several other types of state and federal healthcare fraud and abuse laws have been applied in recent years to restrict certain marketing practices in the pharmaceutical industry. These laws include the U.S. Anti-Kickback Statute, U.S. False Claims Act and similar state laws. Because of the breadth of these laws and the narrowness of the safe harbors, it is possible that some of our business activities could be
| S-38 |
The U.S. Anti-Kickback Statute prohibits, among other things, knowingly and willfully offering, paying, soliciting or receiving remuneration to induce, or in return for, purchasing, leasing, ordering or arranging for the purchase, lease or order of any healthcare item or service reimbursable under Medicare, Medicaid or other federally financed healthcare programs. This statute has been interpreted broadly to apply to arrangements between pharmaceutical manufacturers on the one hand and prescribers, purchasers and formulary managers on the other. Although there are several statutory exemptions and regulatory safe harbors protecting certain common activities from prosecution, the exemptions and safe harbors are drawn narrowly, and practices that involve remuneration intended to induce prescribing, purchasing or recommending may be subject to scrutiny if they do not qualify for an exemption or safe harbor. Our practices may not, in all cases, meet all of the criteria for safe harbor protection from anti-kickback liability. Moreover, recent health care reform legislation has strengthened these laws. For example, the ACA, among other things, amends the intent requirement of the U.S. Anti-Kickback Statute and criminal health care fraud statutes; a person or entity no longer needs to have actual knowledge of this statute or specific intent to violate it. In addition, the ACA provides that the government may assert that a claim including items or services resulting from a violation of the U.S. Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the U.S. False Claims Act. Federal false claims laws prohibit any person from knowingly presenting, or causing to be presented, a false claim for payment to the federal government or knowingly making, or causing to be made, a false statement to get a false claim paid.
Over the past few years, several pharmaceutical and other healthcare companies have been prosecuted under these laws for a variety of alleged promotional and marketing activities, such as: allegedly providing free trips, free goods, sham consulting fees and grants and other monetary benefits to prescribers; reporting to pricing services inflated average wholesale prices that were then used by federal programs to set reimbursement rates; engaging in off-label promotion that caused claims to be submitted to Medicare or Medicaid for non-covered, off-label uses; and submitting inflated best price information to the Medicaid Rebate Program to reduce liability for Medicaid rebates. Most states also have statutes or regulations similar to the U.S. Anti-Kickback Statute and the U.S. False Claims Act, which apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor. Sanctions under these federal and state laws may include substantial civil monetary penalties, exclusion of a manufacturer’s products from reimbursement under government programs, substantial criminal fines and imprisonment.
We have been and expect to be significantly dependent on our collaborative agreements for the development of MAT2203, which exposes us to the risk of reliance on the performance of third parties.
In conducting our research and development activities for MAT2203, we currently rely, and expect to continue to rely, on collaborative agreements with universities, governmental agencies and not-for-profit organizations for both strategic and financial resources. Key among these agreements is our collaboration agreements with the NIH for the development of MAT2203. The loss of, or failure to perform by us or our partners under any applicable agreements or arrangements, or our failure to secure additional agreements for our product candidates, would substantially disrupt or delay our research and development activities, including our in-process and anticipated clinical trials. Any such loss would likely increase our expenses and materially harm our business, financial condition and results of operation.
We expect that we will rely on third parties to conduct clinical trials for our product candidates. If these third parties do not successfully carry out their contractual duties or meet expected deadlines, we may not be able to obtain regulatory approval for or commercialize MAT9001, MAT2203 or any other product candidates that we may develop and our business could be substantially harmed.
We expect to enter into agreements with third-party CROs, or governmental entities like the NIH, to conduct and manage our clinical programs. We rely heavily on these parties for execution of clinical studies for MAT9001, and MAT2203 and our other product candidates and can control only certain and very limited aspects of their activities. Nevertheless, we would be responsible for ensuring that each of our studies is conducted in accordance with the applicable protocol, legal, regulatory and scientific standards, and our reliance on the NIH or CROs would not relieve us of our regulatory responsibilities. We, the NIH and our CROs would be required to comply with cGCPs, which are regulations and guidelines enforced by the FDA, the Competent Authorities of the Member States of the European Economic Area and comparable foreign regulatory authorities for any products in clinical development. The FDA enforces these cGCP regulations through periodic inspections of trial sponsors, principal investigators and trial sites. If we or the NIH or our CROs fail to comply with applicable cGCPs, the clinical data generated in our clinical trials may be deemed unreliable and the FDA or comparable foreign regulatory authorities may require us to perform additional clinical trials before approving our marketing applications. We cannot assure you that, upon inspection, the FDA will determine that any of our clinical trials comply with cGCPs. In addition, our clinical trials must be conducted with products produced under cGMP regulations and will require a large number of test subjects. Our failure or the failure of the NIH or our CROs to comply with these regulations may require us to repeat clinical trials, which would delay the regulatory approval process and could also subject us to enforcement action up to and including civil and criminal penalties.
| S-39 |
As a result, many important aspects of our drug development programs would be outside of our direct control. In addition, the NIH or the CROs may not perform all of their obligations under their arrangements with us or in compliance with regulatory requirements. If NIH or the CROs do not perform clinical trials in a satisfactory manner, breach their obligations to us or fail to comply with regulatory requirements, the development and commercialization of MAT2203, MAT9001 or any other product candidates that we may develop may be delayed or our development program may be materially and irreversibly harmed. We cannot control the amount and timing of resources these CROs would devote to our program or our product candidates. If we are unable to rely on the clinical data collected by our CROs, we could be required to repeat, extend the duration of, or increase the size of our clinical trials, which could significantly delay commercialization and require significantly greater expenditures. If any of our relationships with these third-party CROs terminate, we may not be able to enter into arrangements with alternative CROs. As a result of the foregoing, our financial results and the commercial prospects for MAT9001, MAT2203 and our other product candidates would be harmed, our costs could increase and our ability to generate revenue could be delayed.
Reimbursement decisions by third-party payors may have an adverse effect on pricing and market acceptance of MAT9001, MAT2203 or any other product candidates that we may develop. If there is not sufficient reimbursement for our future products, it is less likely that such products will be widely used.
Market acceptance and sales of MAT9001, MAT2203 or any other product candidates for which we obtain regulatory approval will depend on reimbursement policies and may be affected by future healthcare reform measures in both the United States and foreign jurisdictions. Government authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which products they will cover and establish payment levels. In addition, government authorities and these third-party payors are increasingly attempting to contain health care costs by demanding price discounts or rebates and limiting both the types and variety of products that they will cover and the amounts that they will pay for these products. In addition, we might need to conduct post-marketing studies in order to demonstrate the cost-effectiveness of any future products to such payors’ satisfaction. Such studies might require us to commit a significant amount of management time and financial and other resources.
Reimbursement rates may vary according to the use of the product and the clinical setting in which it is used, may be based on payments allowed for lower-cost products that are already reimbursed, may be incorporated into existing payments for other products or services, and may reflect budgetary constraints and/or imperfections in Medicare or Medicaid data used to calculate these rates. Net prices for products may be reduced by mandatory discounts or rebates required by government health care programs. Such legislation, or similar regulatory changes or relaxation of laws that restrict imports of products from other countries, could reduce the net price we receive for any future marketed products. As a result, our future products might not ultimately be considered cost-effective. We cannot be certain that reimbursement will be available for MAT9001, MAT2203 or any other product candidates that we develop. Also, we cannot be certain that reimbursement policies will not reduce the demand for, or the price paid for, any future products. If reimbursement is not available or is available on a limited basis, we may not be able to successfully commercialize MAT9001, MAT2203 or any other product candidates that we develop.
| S-40 |
MAT9001 is designed to be a prescription-only omega-3 fatty acid based medication. Omega-3 fatty acid based products are also marketed by other companies as dietary supplements, which, unlike drugs, are not subject to FDA approval and therefore do not require a prescription and are not subject to pharmaceutical manufacturing standards. As a result, MAT9001, if approved, would be subject to competition from products for which no prescription is required.
If approved by the regulatory authorities, MAT9001 will be a prescription-only omega-3 fatty acid-based medication. Mixtures of omega-3 fatty acids are naturally occurring substances in various foods, including fatty fish. Omega-3 fatty acids are also marketed as dietary supplements, which may generally be marketed without a lengthy FDA premarket review and approval process and are not subject to prescription. However, unlike prescription drug products, manufacturers of dietary supplements may not make therapeutic claims for their products; dietary supplements may be marketed with claims describing how the product affects the structure or function of the body without premarket approval, but may not expressly or implicitly represent that the dietary supplement will diagnose, cure, mitigate, treat, or prevent disease. We believe the exact omega-3 fatty acid composition and pharmaceutical-grade purity of MAT9001 has a superior therapeutic profile to the omega-3 compositions in commercially available dietary supplements. However, we cannot be sure that physicians or consumers will view MAT9001 as superior. To the extent the price of MAT9001 is significantly higher than the prices of commercially available omega-3 fatty acids marketed by other companies as dietary supplements, physicians may recommend these commercial alternatives instead of MAT9001 or patients may elect on their own to take commercially available non-prescription omega-3 fatty acids. Either of these outcomes may adversely impact our results of operations by limiting product sales and how we price our product, thereby limiting the revenue we receive from sales of MAT9001
Unfavorable pricing regulations, third-party reimbursement practices or healthcare reform initiatives could harm our business.
There is increasing pressure on biotechnology companies to reduce healthcare costs. In the U.S., these pressures come from a variety of sources, such as managed care groups, institutional, government purchasers and government leaders. For example, President Trump has indicated support for possible new measures related to drug pricing. Increased purchasing power of entities that negotiate on behalf of federal healthcare programs and private sector beneficiaries could increase pricing pressures in the future. Such pressures may also increase the risk of litigation or investigation by the government regarding pricing calculations. The biotechnology industry will likely face greater regulation and political and legal action in the future.
The regulations that govern marketing approvals, pricing and reimbursement for new therapeutic products vary widely from country to country. Some countries require approval of the sale price of a product before it can be marketed. In many countries, the pricing review period begins after marketing or product licensing approval is granted. In some foreign markets, prescription pharmaceutical pricing remains subject to continuing governmental control even after initial approval is granted. As a result, we might obtain regulatory approval for a product in a particular country, but then be subject to price regulations that delay our commercial launch of the product and negatively impact the revenue we are able to generate from the sale of the product in that country.
Adverse pricing limitations may hinder our ability to recoup our investment in one or more product candidates, even if our product candidates obtain regulatory approval. Adverse pricing limitations prior to approval will also adversely affect us by reducing our commercial potential. Our ability to commercialize any products successfully also will depend in part on the extent to which reimbursement for these products and related treatments becomes available from government health administration authorities, private health insurers and other organizations.
Government authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which medications they will pay for and establish reimbursement levels.
A primary trend in the U.S. healthcare industry and elsewhere is cost containment. Government authorities and these third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular medications. Increasingly, third-party payors are requiring that companies provide them with predetermined discounts from list prices and are challenging the prices charged for medical products. We cannot be sure that coverage and reimbursement will be available for any product that we commercialize and, if reimbursement is available, what the level of reimbursement will be. Reimbursement may impact the demand for, or the price of, any product for which we obtain marketing approval. Obtaining reimbursement for our products may be particularly difficult because of the higher prices often associated with products administered under the supervision of a physician. If reimbursement is not available or is available only to limited levels, we may not be able to successfully commercialize any product candidate that we successfully develop.
| S-41 |
There may be significant delays in obtaining reimbursement for approved products, and coverage may be more limited than the purposes for which the product is approved by the FDA or regulatory authorities in other countries. Moreover, eligibility for reimbursement does not imply that any product will be paid for in all cases or at a rate that covers our costs, including research, development, manufacture, sale and distribution. Interim payments for new products, if applicable, may also not be sufficient to cover our costs and may not be made permanent. Payment rates may vary according to the use of the product and the clinical setting in which it is used, may be based on payments allowed for lower cost products that are already reimbursed and may be incorporated into existing payments for other services. Net prices for products may be reduced by mandatory discounts or rebates required by government healthcare programs or private payors and by any future relaxation of laws that presently restrict imports of products from countries where they may be sold at lower prices than in the United States. Third-party payors often rely upon Medicare coverage policy and payment limitations in setting their own reimbursement policies. Our inability to promptly obtain coverage and profitable payment rates from both government funded and private payors for new products that we develop could have a material adverse effect on our operating results, our ability to raise capital needed to commercialize products and our overall financial condition.
We are, and will be, completely dependent on third parties to manufacture MAT9001, and our commercialization of MAT9001 could be halted, delayed or made less profitable if those third parties fail to obtain manufacturing approval from the FDA or comparable foreign regulatory authorities, fail to provide us with sufficient quantities of MAT9001 or fail to do so at acceptable quality levels or prices.
We do not currently have, nor do we plan to acquire, the capability or infrastructure to manufacture the active pharmaceutical ingredient, or API, in MAT9001 for use in our clinical trials or for commercial product, if any. In addition, we do not have the capability to encapsulate MAT9001 as a finished drug product for commercial distribution. As a result, we will rely on contract manufacturers throughout the development process and then if and when MAT9001 is approved for commercialization. We have not entered into any agreement with any contract manufacturers for commercial supply and may not be able to engage a contract manufacturer for commercial supply of MAT9001 on favorable terms to us, or at all.
The facilities used by our contract manufacturers to manufacture MAT9001 must be approved by the FDA pursuant to inspections that will be conducted after we submit our NDA to the FDA. We do not control the manufacturing process of, and are completely dependent on, our contract manufacturing partners for compliance with cGMPs for manufacture of both active drug substances and finished drug products. These cGMP regulations cover all aspects of the manufacturing, testing, quality control and record keeping relating to MAT9001. If our contract manufacturers cannot successfully manufacture material that conforms to our specifications and the strict regulatory requirements of the FDA or others, they will not be able to secure and/or maintain regulatory approval for their manufacturing facilities. If the FDA or a comparable foreign regulatory authority does not approve these facilities for the manufacture of MAT9001 or if it withdraws any such approval in the future, we may need to find alternative manufacturing facilities, which would significantly impact our ability to develop, obtain regulatory approval for or market MAT9001, if approved.
Our contract manufacturers will be subject to ongoing periodic unannounced inspections by the FDA and corresponding state and foreign agencies for compliance with cGMPs and similar regulatory requirements. We do not have control over our contract manufacturers’ compliance with these regulations and standards. Failure by any of our contract manufacturers to comply with applicable regulations could result in sanctions being imposed on us, including fines, injunctions, civil penalties, failure to grant approval to market MAT9001, delays, suspensions or withdrawals of approvals, operating restrictions and criminal prosecutions, any of which could significantly and adversely affect our business. In addition, we have no control over the ability of our contract manufacturers to maintain adequate quality control, quality assurance and qualified personnel. Failure by our contract manufacturers to comply with or maintain any of these standards could adversely affect our ability to develop, obtain regulatory approval for or market MAT9001.
If, for any reason, these third parties are unable or unwilling to perform, we may not be able to terminate our agreements with them, and we may not be able to locate alternative manufacturers or formulators or enter into favorable agreements with them and we cannot be certain that any such third parties will have the manufacturing capacity to meet future requirements. If these manufacturers or any alternate manufacturer of finished drug product experiences any significant difficulties in its respective manufacturing processes for our API or finished MAT9001 product or should cease doing business with us, we could experience significant interruptions in the supply of MAT9001 or may not be able to create a supply of MAT9001 at all. Were we to encounter manufacturing issues, our ability to produce a sufficient supply of MAT9001 might be negatively affected. Our inability to coordinate the efforts of our third party manufacturing partners, or the lack of capacity available at our third party manufacturing partners, could impair our ability to supply MAT9001 at required levels. Because of the significant regulatory requirements that we would need to satisfy in order to qualify a new bulk or finished product manufacturer, if we face these or other difficulties with our current manufacturing partners, we could experience significant interruptions in the supply of MAT9001 if we decided to transfer the manufacture of MAT9001 to one or more alternative manufacturers in an effort to deal with the difficulties.
| S-42 |
Any manufacturing problem or the loss of a contract manufacturer could be disruptive to our operations and result in lost sales. Additionally, we rely on third parties to supply the raw materials needed to manufacture our potential products. Any reliance on suppliers may involve several risks, including a potential inability to obtain critical materials and reduced control over production costs, delivery schedules, reliability and quality. Any unanticipated disruption to a future contract manufacturer caused by problems at suppliers could delay shipment of MAT9001, increase our cost of goods sold and result in lost sales.
We cannot guarantee that our manufacturing and supply partners will be able to reduce the costs of commercial scale manufacturing of MAT9001 over time. If the commercial-scale manufacturing costs of MAT9001 are higher than expected, these costs may significantly impact our operating results. In order to reduce costs, we may need to develop and implement process improvements. However, in order to do so, we will need, from time to time, to notify or make submissions with regulatory authorities, and the improvements may be subject to approval by such regulatory authorities. We cannot be sure that we will receive these necessary approvals or that these approvals will be granted in a timely fashion. We also cannot guarantee that we will be able to enhance and optimize output in our commercial manufacturing process. If we cannot enhance and optimize output, we may not be able to reduce our costs over time.
Risks Relating to Our Intellectual Property Rights and Regulatory Exclusivity
We depend on certain technologies that are licensed to us. We do not control these technologies and any loss of our rights to them could prevent us from discovering, developing and commercializing our product candidates.
We rely partially upon our LNC platform delivery technology which is licensed to us by Rutgers. We do not own the patents that underlie this technology. Our rights to use the technology we license are subject to the negotiation of, continuation of and compliance with the terms of our license agreement with Rutgers. Pursuant to the terms of our license agreement with Rutgers, we control the prosecution, maintenance, or filing of the patents to which we hold licenses, as well as the enforcement of these patents against third parties. However, some of our patents and patent applications were either acquired from another company who acquired those patents and patent applications from yet another company, or are licensed from a third party. Thus, these patents and patent applications were not written by us or our attorneys, and we did not have control over the drafting and prosecution of certain of these patents. The former patent owners and our licensors might not have given the same attention to the drafting and prosecution of these patents and applications as we would have if we had been the owners of the patents and applications and had control over the drafting and prosecution. We cannot be certain that drafting and/or prosecution of the licensed patents and patent applications by the licensors have been or will be conducted in compliance with applicable laws and regulations or will result in valid and enforceable patents and other intellectual property rights.
Our rights to use the technology we license are subject to the validity of the owner’s intellectual property rights. Enforcement of our licensed patents or defense or any claims asserting the invalidity of these patents is often subject to the control or cooperation of our licensors. Legal action could be initiated against the owners of the intellectual property that we license and an adverse outcome in such legal action could harm our business because it might prevent such companies or institutions from continuing to license intellectual property that we may need to operate our business. In addition, such licensors may resolve such litigation in a way that benefits them but adversely affects our ability to use the licensed technology for our products.
Certain of our licenses contained in our agreement with Rutgers contain provisions that allow the licensor to terminate the license if (i) we breach any payment obligation or other material provision under the agreement and fail to cure the breach within a fixed time following written notice of termination, (ii) we or any of our affiliates, licensees or sub licensees directly or indirectly challenge the validity, enforceability, or extension of any of the licensed patents or (iii) we declare bankruptcy or dissolve. Our rights under the licenses are subject to our continued compliance with the terms of the license, including the payment of royalties due under the license. Termination of these licenses would prevent us from discovering, developing and commercializing product candidates based on the cochleate delivery technology, including our lead anti-infective product candidate, MAT2203. Determining the scope of the license and related royalty obligations can be difficult and can lead to disputes between us and the licensor. An unfavorable resolution of such a dispute could lead to an increase in the royalties’ payable pursuant to the license. If a licensor believed we were not paying the royalties due under the license or were otherwise not in compliance with the terms of the license, the licensor might attempt to revoke the license. If such an attempt were successful, we might be barred from discovering, developing and commercializing product candidates based on the cochleate delivery technology, including our lead anti-infective product candidate.
| S-43 |
If we discontinue development of the cochleate delivery technology, we would be required to return such technology to the former stockholders of Aquarius and we would lose the rights to our lead product candidates.
Under certain circumstances, we will be required to transfer Aquarius’ cochleate delivery technology back to the former shareholders of Aquarius. This transfer would be required under the Merger Agreement in the event the following conditions are met: (i) no milestone events have occurred on or before the two-year anniversary of the effective time of the Aquarius Merger (the “Transfer Date”), (ii) during such period we shall have discontinued efforts to develop or commercialize the cochleate delivery technology (as conclusively demonstrated by our omission of the cochleate delivery technology in at least two consecutive royalty, progress and payment reports delivered to Rutgers pursuant to the license agreement entered into between Aquarius and Rutgers) and (iii) as of the Transfer Date, no unresolved indemnification claims for us and our indemnified parties are pending. If the foregoing conditions are met, we would transfer the cochleate delivery technology to the stockholder representative or to a newly formed entity as directed by the stockholder representative (in either case for the benefit of the former Aquarius stockholders) following receipt of any necessary third party consents required for the transfer, which we shall use its commercially reasonable efforts to obtain. If we are required to transfer the cochleate delivery technology back to the former shareholders of Aquarius, we would lose our rights to our lead product candidates, which would have a material and adverse effect on our business.
It is difficult and costly to protect our intellectual property rights, and we cannot ensure the protection of these rights.
Our commercial success will depend, in part, on obtaining and maintaining patent protection for our technologies, products and processes, successfully defending these patents against third-party challenges and successfully enforcing these patents against third party competitors. The patent positions of pharmaceutical companies can be highly uncertain and involve complex legal, scientific and factual questions for which important legal principles remain unresolved. Changes in either the patent laws or in interpretations of patent laws may diminish the value of our intellectual property. Accordingly, we cannot predict the breadth of claims that may be allowable or enforceable in our patents (including patents owned and licensed by us). We currently own or have rights to 22 issued patents relating to our cochleate delivery technology, as well as pending patent applications for our cochleate delivery technology that may never be approved by the United States or foreign patent offices. Furthermore, any patents which may eventually be issued from existing patent applications for any of our technologies, may be challenged, invalidated or circumvented by third parties and might not protect us against competitors with similar products or technologies.
The degree of future protection for our proprietary rights is uncertain, because legal means afford only limited protection and may not adequately protect our rights, permit us to gain or keep our competitive advantage, or provide us with any competitive advantage at all. We cannot be certain that any patent application owned by a third party will not have priority over patent applications filed by us, or that we will not be involved in interference, opposition or invalidity proceedings before the United States or foreign patent offices.
We also rely on trade secrets to protect technology, especially in cases where we believe patent protection is not appropriate or obtainable. However, trade secrets are difficult to protect. While we require employees, academic collaborators, consultants and other contractors to enter into confidentiality agreements, we may not be able to adequately protect our trade secrets or other proprietary or licensed information. Typically, research collaborators and scientific advisors have rights to publish data and information in which we may have rights. If we cannot maintain the confidentiality of our proprietary technology and other confidential information, our ability to receive patent protection and our ability to protect valuable information owned by us may be imperiled. Enforcing a claim that a third-party entity illegally obtained and is using any of our trade secrets is expensive and time consuming, and the outcome is unpredictable. In addition, courts are sometimes less willing to protect trade secrets than patents. Moreover, our competitors may independently develop equivalent knowledge, methods and know-how.
| S-44 |
If we fail to obtain or maintain patent protection or trade secret protection for our technologies, third parties could use our proprietary information, which could impair our ability to compete in the market and adversely affect our ability to generate revenues and attain profitability.
We may also develop trademarks to distinguish our products from the products of our competitors. We cannot guarantee that any trademark applications filed by us or our business partners will be approved. Third parties may also oppose such trademark applications, or otherwise challenge our use of the trademarks. In the event that the trademarks we use are successfully challenged, we could be forced to rebrand our products, which could result in loss of brand recognition, and could require us to devote resources to advertising and marketing new brands. Further, we cannot provide assurance that competitors will not infringe the trademarks we use, or that we will have adequate resources to enforce these trademarks.
Our product candidates may infringe the intellectual property rights of others, which could increase our costs and delay or prevent our development and commercialization efforts.
Our success depends in part on avoiding infringement of the proprietary technologies of others. The pharmaceutical industry has been characterized by frequent litigation regarding patent and other intellectual property rights. Identification of third party patent rights that may be relevant to our proprietary technology is difficult because patent searching is imperfect due to differences in terminology among patents, incomplete databases and the difficulty in assessing the meaning of patent claims. Additionally, because patent applications are maintained in secrecy until the application is published, we may be unaware of third-party patents that may be infringed by commercialization of MAT9001, MAT2203 or any future product candidate. There may be certain issued patents and patent applications claiming subject matter that we may be required to license in order to research, develop or commercialize MAT9001 or MAT2203 and we do not know if such patents and patent applications would be available to license on commercially reasonable terms, or at all. Any claims of patent infringement asserted by third parties against us would be time-consuming and may:
| ● | result in costly litigation; | |
| ● | divert the time and attention of our technical personnel and management; | |
| ● | prevent us from commercializing a product until the asserted patent expires or is held finally invalid or not infringed in a court of law; | |
| ● | require us to cease or modify our use of the technology and/or develop non-infringing technology; or | |
| ● | require us to enter into royalty or licensing agreements. |
Although no third party has asserted a claim of infringement against us, others may hold proprietary rights that could prevent MAT9001 or MAT2203 from being marketed. Any patent-related legal action against us claiming damages and seeking to enjoin commercial activities relating to MAT9001 or MAT2203 or our processes could subject us to potential liability for damages and require us to obtain a license to continue to manufacture or market our current product candidates or any future product candidates. We cannot predict whether we would prevail in any such actions or that any license required under any of these patents would be made available on commercially acceptable terms, if at all. In addition, we cannot be sure that we could redesign, MAT9001, MAT2203, or any future product candidates or processes to avoid infringement, if necessary. Accordingly, an adverse determination in a judicial or administrative proceeding, or the failure to obtain necessary licenses, could prevent us from developing and commercializing MAT9001, MAT2203 or a future product candidate, which could harm our business, financial condition and operating results.
We anticipate that competitors may from time to time oppose our efforts to obtain patent protection for new technologies or to submit patented technologies for regulatory approval. Competitors may seek to oppose our patent applications to delay the approval process or to challenge our granted patents, for example, by requesting a reexamination of our patent at the United States Patent and Trademark Office, or the USPTO, or by filing an opposition in a foreign patent office, even if the opposition or challenge has little or no merit. Such proceedings are generally highly technical, expensive and time consuming, and there can be no assurance that such a challenge would not result in the narrowing or complete revocation of any patent of ours that was so challenged.
| S-45 |
We may be subject to claims that we have wrongfully hired an employee from a competitor or that we or our employees have wrongfully used or disclosed alleged confidential information or trade secrets of their former employers.
As is commonplace in our industry, we employ individuals who were previously employed at or retained by other pharmaceutical companies, including our competitors or potential competitors. Although no claims against us are currently pending, we may be subject in the future to claims that our employees or prospective employees are subject to a continuing obligation to their former employers (such as non-competition or non-solicitation obligations) or claims that our employees or we have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of their former employers. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to management.
We may not be able to obtain or maintain orphan drug designation or exclusivity for our anti-infective product candidates.
We may seek orphan drug designation for MAT2203 in the United States and may seek additional orphan drug designation for other product candidates. Regulatory authorities in some jurisdictions, including the United States and Europe, may designate drugs for relatively small patient populations as orphan drugs. Under the Orphan Drug Act, the FDA may designate a product as an orphan drug if it is a drug intended to treat a rare disease or condition, which is generally defined as a patient population of fewer than 200,000 individuals in the United States. Generally, if a product with an orphan drug designation subsequently receives the first marketing approval for the indication for which it has such designation, the product is entitled to a period of marketing exclusivity, which precludes the FDA or the EMA from approving another marketing application for the same indication for that drug during that time period. For a product that obtains orphan drug designation on the basis of a plausible hypothesis that it is clinically superior to the same drug that is already approved for the same indication, in order to obtain orphan drug exclusivity upon approval, clinical superiority of such product to this same drug that is already approved for the same orphan indication must be demonstrated. The exclusivity period is seven years in the United States and ten years in Europe. The European exclusivity period can be reduced to six years if a drug no longer meets the criteria for orphan drug designation or if the drug is sufficiently profitable so that market exclusivity is no longer justified. Orphan drug exclusivity may be lost if the FDA or the EMA determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantity of the drug to meet the needs of patients with the rare disease or condition.
We cannot assure you that the application for orphan drug designation of MAT2203, or any future application with respect to any other product candidate, will be granted. If we are unable to obtain orphan drug designation in the United States, we will not be eligible to obtain the period of market exclusivity that could result from orphan drug designation or be afforded the financial incentives associated with orphan drug designation. Even if we obtain orphan drug exclusivity for a product, that exclusivity may not effectively protect the product from competition because different drugs can be approved for the same condition. Even after an orphan drug is approved, the FDA can subsequently approve the same drug for the same condition if the FDA concludes that the later drug is clinically superior in that it is shown to be safer, more effective or makes a major contribution to patient care.
| S-46 |
Any fast track designation or grant of priority review status by the FDA may not actually lead to a faster development or regulatory review or approval process, nor will it assure FDA approval of our product candidates. Additionally, our product candidates may treat indications that do not qualify for priority review vouchers.
We have received fast track designation for MAT2203 for the treatment of invasive candidiasis, the treatment of aspergillosis and the prevention of invasive fungal infections due to immunosuppressive therapy and may seek fast track designation for some of our other product candidates or priority review of applications for approval of our product candidates for certain indications. If a drug is intended for the treatment of a serious or life-threatening condition and the drug demonstrates the potential to address unmet medical needs for this condition, the drug sponsor may apply for FDA fast track designation. If a product candidate offers major advances in treatment, the FDA may designate it eligible for priority review. The FDA has broad discretion whether or not to grant these designations, so even if we believe a particular product candidate is eligible for these designations, we cannot assure you that the FDA would decide to grant them. Even if we do receive fast track designation or priority review, we may not experience a faster development process, review or approval compared to conventional FDA procedures. The FDA may withdraw fast track designation if it believes that the designation is no longer supported by data from our clinical development program.
Any breakthrough therapy designation granted by the FDA for our product candidates may not lead to a faster development or regulatory review or approval process, and it does not increase the likelihood that our product candidates will receive marketing approval.
We may seek a breakthrough therapy designation for some of our product candidates. A breakthrough therapy is defined as a drug that is intended, alone or in combination with one or more other drugs, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. For drugs and biologics that have been designated as breakthrough therapies, interaction and communication between the FDA and the sponsor of the trial can help to identify the most efficient path for clinical development while minimizing the number of patients placed in ineffective control regimens. Drugs designated as breakthrough therapies by the FDA may also be eligible for accelerated approval if the relevant criteria are met.
Designation as a breakthrough therapy is within the discretion of the FDA. Accordingly, even if we believe one of our product candidates meets the criteria for designation as a breakthrough therapy, the FDA may disagree and instead determine not to make such designation. In any event, the receipt of a breakthrough therapy designation for a product candidate may not result in a faster development process, review or approval compared to drugs considered for approval under conventional FDA procedures and does not assure ultimate approval by the FDA. In addition, even if one or more of our product candidates qualify as breakthrough therapies, the FDA may later decide that the products no longer meet the conditions for qualification or decide that the time period for FDA review or approval will not be shortened.
Designation of our product candidates as qualified infectious disease products is not assured and, in any event, even if granted, may not actually lead to a faster development or regulatory review, and would not assure FDA approval of our product candidates.
We have received a qualified infectious disease product, or QIDP, designation for MAT2203 for certain indications and we may be eligible for designation of certain of our product candidates as QIDPs. A QIDP is “an antibacterial or antifungal drug intended to treat serious or life-threatening infections, including those caused by an antibacterial or antifungal resistant pathogen, including novel or emerging infectious pathogens or certain “qualifying pathogens.” A product designated as a QIDP will be granted priority review by the FDA and may qualify for “fast track” status. Upon the approval of an NDA for a drug product designated by the FDA as a QIDP, the product is granted a period of five years of regulatory exclusivity in addition to any other period of regulatory exclusivity for which the product is eligible. The FDA has broad discretion whether or not to grant these designations, so even if we believe a particular product candidate is eligible for such designation or status, the FDA could decide not to grant it. Moreover, even if we do receive such a designation, we may not experience a faster development process, review or approval compared to conventional FDA procedures and there is no assurance that our product candidate, even if determined to be a QIDP, will be approved by the FDA.
| S-47 |
General Company-Related Risks
We will need to increase the size of our organization to grow our business, and we may experience difficulties in managing this growth.
We currently have only fifteen employees. As our development and commercialization plans and strategies develop, we will need to expand the size of our employee base for managerial, development, operational, sales, marketing, financial and other resources. Future growth would impose significant added responsibilities on members of management, including the need to identify, recruit, maintain, motivate and integrate additional employees. In addition, our management may have to divert a disproportionate amount of its attention away from our day-to-day activities and devote a substantial amount of time to managing these growth activities. Our future financial performance and our ability to commercialize our product candidates and our ability to compete effectively will depend, in part, on our ability to effectively manage any future growth.
If we are not successful in attracting and retaining highly qualified personnel, we may not be able to successfully implement our business strategy. In addition, the loss of the services of certain key employees would adversely impact our business prospects.
If we are not successful in attracting and retaining highly qualified personnel, we may not be able to successfully implement our business strategy. In addition, the loss of the services of certain key employees, including Jerome D. Jabbour, our Chief Executive Officer and President, would adversely impact our business prospects.
Our ability to compete in the highly competitive pharmaceutical industry depends in large part upon our ability to attract highly qualified managerial, scientific and medical personnel. In order to induce valuable employees to remain with us, we intend to provide employees with stock options that vest over time. The value to employees of stock options that vest over time will be significantly affected by movements in our stock price that we will not be able to control and may at any time be insufficient to counteract more lucrative offers from other companies.
Other pharmaceutical companies with which we compete for qualified personnel have greater financial and other resources, different risk profiles, and a longer history in the industry than we do. They also may provide more diverse opportunities and better chances for career advancement. Some of these characteristics may be more appealing to high-quality candidates than what we have to offer. If we are unable to continue to attract and retain high-quality personnel, the rate and success at which we can develop and commercialize product candidates would be limited.
If product liability lawsuits are brought against us, we may incur substantial liabilities and may be required to limit commercialization of our product candidates.
We face a potential risk of product liability as a result of the clinical testing of MAT9001, MAT2203 or any future product candidates and will face an even greater risk if we commercialize MAT9001, MAT2203 or any other future product. For example, we may be sued if any product we develop or any material that we use in our products allegedly causes injury or is found to be otherwise unsuitable during product testing, manufacturing, marketing or sale. Any such product liability claims may include allegations of defects in manufacturing, defects in design, a failure to warn of dangers inherent in the product, negligence, strict liability and a breach of warranties. Claims could also be asserted under state consumer protection acts. If we cannot successfully defend ourselves against product liability claims, we may incur substantial liabilities or be required to limit commercialization of MAT9001 or MAT2203. Even successful defense would require significant financial and management resources. Regardless of the merits or eventual outcome, liability claims may result in:
| ● | decreased demand for MAT9001, MAT2203 or any future products that we may develop; | |
| ● | injury to our reputation; | |
| ● | withdrawal of clinical trial participants; | |
| ● | costs to defend the related litigation; | |
| ● | a diversion of management’s time and our resources; | |
| ● | substantial monetary awards to trial participants or patients; | |
| ● | product recalls, withdrawals or labeling, marketing or promotional restrictions; | |
| ● | loss of revenue; | |
| ● | the inability to commercialize our product candidates; and | |
| ● | a decline in our stock price. |
| S-48 |
Our inability to obtain and retain sufficient product liability insurance at an acceptable cost to protect against potential product liability claims could prevent or inhibit the commercialization of products we develop. We have obtained product liability insurance covering our clinical trials in the amount of greater than or equal to $5 million in the aggregate. Although we will maintain such insurance, any claim that may be brought against us could result in a court judgment or settlement in an amount that is not covered, in whole or in part, by our insurance or that is in excess of the limits of our insurance coverage. Our insurance policies also have various exclusions, and we may be subject to a product liability claim for which we have no coverage. We may have to pay any amounts awarded by a court or negotiated in a settlement that exceed our coverage limitations or that are not covered by our insurance, and we may not have, or be able to obtain, sufficient capital to pay such amounts.
Our internal computer systems, or those of our CROs or other contractors or consultants, may fail or suffer security breaches, which could result in a material disruption of our product development programs.
Despite the implementation of security measures, our internal computer systems and those of our CROs and other contractors and consultants are vulnerable to damage or disruption from computer viruses, software bugs, unauthorized access, natural disasters, terrorism, war, and telecommunication, equipment and electrical failures. While we have not, to our knowledge, experienced any significant system failure, accident or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our programs. For example, the loss of clinical trial data from completed or ongoing clinical trials for any of our product candidates could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach results in a loss of or damage to our data or applications, or inappropriate disclosure or theft of confidential or proprietary information, we could incur liability, the further development of our product candidates could be delayed, our competitive position could be compromised, or our business reputation could be harmed.
We may acquire businesses or products, or form strategic alliances, in the future, and we may not realize the benefits of such acquisitions.
We may acquire additional businesses or products, form strategic alliances or create joint ventures with third parties that we believe will complement or augment our existing business. If we acquire businesses with promising markets or technologies, we may not be able to realize the benefit of acquiring such businesses if we are unable to successfully integrate them with our existing operations and company culture. We may encounter numerous difficulties in developing, manufacturing and marketing any new products resulting from a strategic alliance or acquisition that delay or prevent us from realizing their expected benefits or enhancing our business. We cannot assure you that, following any such acquisition, we will achieve the expected synergies to justify the transaction.
Risks related to our Securities
Pursuant to the terms of our outstanding Series A Preferred Stock, we may be obligated to pay significant royalties.
Pursuant to the terms of the Certificate of Designations of Preferences, Rights and Limitations (the “Certificate of Designations”) for our outstanding Series A Preferred Stock, we may be required to pay royalties of up to $35 million per year. If and when we obtain FDA or EMA approval of MAT2203 and/or MAT2501, which we do not expect to occur before 2020, if ever, and/or if we generate sales of such products, or we receive any proceeds from the licensing or other disposition of MAT2203 or MAT2501, we are required to pay to the holders of our Series A Preferred Stock, subject to certain vesting requirements, in aggregate, a royalty equal to (i) 4.5% of Net Sales (as defined in the Certificate of Designations), subject in all cases to a cap of $25 million per calendar year, and (ii) 7.5% of Licensing Proceeds (as defined in the Certificate of Designations), subject in all cases to a cap of $10 million per calendar year. The Royalty Payment Rights will expire when the patents covering the applicable product expire, which is currently expected to be in 2033.
| S-49 |
We are obligated to pay dividends on outstanding shares of our preferred stock.
Holders of Series A Preferred Stock are entitled to receive cumulative dividends at the rate per share of 8% per annum, payable in shares of our common stock, which annual dividend will accumulate until such time as the shares of Series A Preferred Stock are converted, at which time the accumulated dividend will be satisfied by delivery of shares of common stock at a price per share of common stock equal to the conversion price of the Series A Preferred Stock then in effect (currently $0.50 per share). The Series A Preferred Stock will automatically convert at the conversion price in effect on July 29, 2019, unless such shares are converted earlier in accordance with the terms of the Certificate of Designations for the Series A Preferred Stock.
Holders of our Series B Preferred Stock will be entitled to receive dividends payable as follows: (i) a number of shares of common stock equal to 10% of the shares of common stock underlying the Series B Preferred Stock then held by the holder on June 19, 2019, (ii) a number of shares of common stock equal to 15% of the shares of common stock underlying the Series B Preferred then held by such holder on June 19, 2020 and (iii) a number of shares of common stock equal to 20% of the shares of common stock underlying the Series B Preferred then held by such holder on June 19, 2021.
The payment of such dividends will result in additional dilution to our holders of our common stock.
Our outstanding shares of preferred stock have certain preference rights upon any liquidation, dissolution or winding up.
Upon any dissolution, liquidation or winding up, whether voluntary or involuntary, holders of Series A Preferred Stock will be entitled to (i) first receive distributions out of our assets in an amount per share equal to $5.00, or the stated value, plus all accrued and unpaid dividends, whether capital or surplus before any distributions shall be made on any shares of common stock and (ii) second, receive distributions out of our assets on an as-converted basis alongside the common stock.
The rights of the holders of common stock may be impaired by the potential issuance of preferred stock.
Our articles of incorporation give our board of directors the ability to designate and issue preferred stock in one or more series. As a result, the board of directors may, without stockholder approval, issue new series of preferred stock with voting, dividend, conversion, liquidation or other rights which could adversely affect the relative voting power and equity interest of the holders of common stock. Additional issuances of preferred stock, which could be issued with the right to more than one vote per share, could have the effect of discouraging, delaying or preventing a change of control of us. The possible impact on takeover attempts could adversely affect the price of our common stock. Although we have no present intention to designate any new series, or issue any shares, of preferred stock, we may do so in the future.
We do not intend to pay dividends on our common stock in the foreseeable future.
The Board of Directors will determine, in its sole discretion, our dividend policy after considering our financial condition, results of operations and capital requirements, as well as other factors. No dividends may be declared or paid on our common stock, unless a dividend, payable in the same consideration or manner, is simultaneously declared or paid, as the case may be, on the shares of Series A Preferred Stock. We do not anticipate paying cash dividends on our common stock in the foreseeable future and you should not invest in us with the anticipation of receiving dividend income.
An active public trading market for our common stock may not be sustained.
Our common stock was listed on the NYSE American under the symbol “MTNB” on March 2, 2017. Prior to March 2, 2017, our common stock was available for quotation on the OTCQB under the symbol “MTNB.” We cannot assure you that an active trading market will be sustained. A lack of an active market may impair your ability to sell shares of our common stock at the time you wish to sell them or at a price that you consider reasonable. The lack of an active market may also reduce the price of shares of our common stock. An inactive market may also impair our ability to raise capital by selling shares of capital stock and may impair our ability to acquire other companies or technologies by using our common stock as consideration.
| S-50 |
Our share price has been and could remain volatile.
The market price of our common stock has historically experienced and may continue to experience significant volatility. From January 1, 2016 through March 13, 2019, the market price of our common stock has fluctuated from a high of $3.99 per share in the first quarter of 2017 to a low of $0.32 per share in the second quarter of 2018. Our progress in developing our product candidates, the impact of government regulations on our products and industry, the potential sale of a large volume of our common stock by stockholders, our quarterly operating results, changes in general conditions in the economy or the financial markets and other developments affecting us or our competitors could cause the market price of our common stock to fluctuate substantially with significant market losses. If our stockholders sell a substantial number of shares of common stock, especially if those sales are made during a short period of time, those sales could adversely affect the market price of our common stock and could impair our ability to raise capital. In addition, in recent years, the stock market has experienced significant price and volume fluctuations. This volatility has affected the market prices of securities issued by many companies for reasons unrelated to their operating performance and may adversely affect the price of our common stock. In addition, we could be subject to a securities class action litigation as a result of volatility in the price of our stock, which could result in substantial costs and diversion of management’s attention and resources and could harm our stock price, business, prospects, results of operations and financial condition.
An active trading market for our common stock may not be sustained.
Although our common stock is listed on the NYSE, the market for our shares has demonstrated varying levels of trading activity. Furthermore, the current level of trading may not be sustained in the future. The lack of an active market for our common stock may impair investors’ ability to sell their shares at the time they wish to sell them or at a price that they consider reasonable, may reduce the fair market value of their shares and may impair our ability to raise capital to continue to fund operations by selling shares and may impair our ability to acquire additional intellectual property assets by using our shares as consideration.
We do not anticipate paying dividends on our common stock and, accordingly, stockholders must rely on stock appreciation for any return on their investment.
We have never declared or paid cash dividends on our common stock and do not expect to do so in the foreseeable future. The declaration of dividends is subject to the discretion of our board of directors and limitations under applicable law, and will depend on various factors, including our operating results, financial condition, future prospects and any other factors deemed relevant by our board of directors. You should not rely on an investment in our company if you require dividend income from your investment in our company. The success of your investment will likely depend entirely upon any future appreciation of the market price of our common stock, which is uncertain and unpredictable. There is no guarantee that our common stock will appreciate in value.
If securities or industry analysts do not publish research or reports about our business, or if they change their recommendations regarding our stock adversely, our stock price and trading volume could decline.
The trading market for our common stock will be influenced by the research and reports that industry or securities analysts publish about us or our business. Our research coverage by industry and financial analysts is currently limited. Even if our analyst coverage increases, if one or more of the analysts who cover us downgrade our stock, our stock price would likely decline. If one or more of these analysts cease coverage of our company or fail to regularly publish reports on us, we could lose visibility in the financial markets, which in turn could cause our stock price or trading volume to decline.
| S-51 |
We are an “emerging growth company,” and we intend to take advantage of reduced disclosure requirements applicable to “emerging growth companies,” which could make our common stock less attractive to investors.
We are an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act of 2012, or JOBS Act, and, for as long as we continue to be an “emerging growth company,” we intend to take advantage of certain exemptions from various reporting requirements applicable to other public companies but not to “emerging growth companies,” including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. We could be an “emerging growth company” until December 31, 2019, or until the earliest of (i) the last day of the first fiscal year in which our annual gross revenues exceed $1 billion, (ii) the date that we become a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act, which would occur if the market value of our common stock that is held by non-affiliates exceeds $700 million as of the last business day of our most recently completed second fiscal quarter, or (iii) the date on which we have issued more than $1 billion in non-convertible debt during the preceding three year period. We cannot predict if investors will find our common stock less attractive if we choose to continue to rely on these exemptions. If some investors find our common stock less attractive as a result of any choices to reduce future disclosure, there may be a less active trading market for our common stock and our stock price may be more volatile.
We are incurring significantly increased costs and devote substantial management time as a result of operating as a public company, which costs may increase after we are no longer an “emerging growth company.”
As a public company, we are incurring significant legal, accounting and other expenses. For example, we are required to comply with certain of the requirements of the Sarbanes-Oxley Act and the Dodd-Frank Wall Street Reform and Consumer Protection Act, as well as rules and regulations subsequently implemented by the SEC, including the establishment and maintenance of effective disclosure and financial controls and changes in corporate governance practices. Compliance with these requirements have resulted in increased legal and financial compliance costs. In addition, our management and other personnel must divert attention from operational and other business matters to devote substantial time to these public company requirements. In particular, we are incurring significant expenses and devote substantial management effort toward ensuring compliance with the requirements of Section 404 of the Sarbanes-Oxley Act.
However, for as long as we remain an “emerging growth company” as defined in the JOBS Act, we intend to take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not “emerging growth companies” including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. We intend to take advantage of these reporting exemptions until we are no longer an “emerging growth company.”
Under the JOBS Act, “emerging growth companies” can delay adopting new or revised accounting standards until such time as those standards apply to private companies. We have irrevocably elected not to avail ourselves of this exemption from new or revised accounting standards and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not “emerging growth companies.”
After we are no longer an “emerging growth company” we expect to incur additional management time and cost to comply with the more stringent reporting requirements applicable to companies that are deemed accelerated filers or large accelerated filers, including complying with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act.
We cannot predict or estimate the amount of additional costs we may incur as a result of becoming a public company or the timing of such costs.
We identified a material weakness in our internal control over financial reporting. If we are not able to remediate the material weakness and otherwise maintain an effective system of internal control over financial reporting, the reliability of our financial reporting, investor confidence in us and the value of our common stock could be adversely affected.
| S-52 |
As a public company, we are required to maintain internal control over financial reporting and to report any material weaknesses in such internal controls. Section 404 of SOX, or Section 404, requires that we evaluate and determine the effectiveness of our internal controls over financial reporting and provide a management report on internal control over financial reporting. A material weakness is a deficiency, or combination of deficiencies, in internal control over financial reporting such that there is a reasonable possibility that a material misstatement of annual or interim financial statements will not be prevented or detected and corrected on a timely basis.
In connection with the audits of fiscal year 2017 financial statement, we identified a material weakness in our internal control over financial reporting related to our controls over accounting for stock-based compensation, which allowed for the misinterpretation and historical misapplication of Accounting Standards Codification (“ASC”) 718, Compensation – Stock compensation, regarding the modification of stock option awards issued to employees. To remediate the material weakness described above, we have initiated compensating controls in the near term and are enhancing and revising the design of existing controls and procedures to properly apply ASC 718 to the cancellation and replacement of stock-based compensation awards.
If our steps are insufficient to successfully remediate the material weakness and otherwise establish and maintain an effective system of internal control over financial reporting, the reliability of our financial reporting, investor confidence in us and the value of our common stock could be materially and adversely affected. Effective internal control over financial reporting is necessary for us to provide reliable and timely financial reports and, together with adequate disclosure controls and procedures, are designed to reasonably detect and prevent fraud. Any failure to implement required new or improved controls, or difficulties encountered in their implementation could cause us to fail to meet our reporting obligations. For as long as we are an “emerging growth company” under the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, our independent registered public accounting firm will not be required to attest to the effectiveness of our internal control over financial reporting pursuant to Section 404. We could be an “emerging growth company” until December 31, 2019, or until the earliest of (i) the last day of the first fiscal year in which our annual gross revenues exceed $1 billion, (ii) the date that we become a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act, which would occur if the market value of our common stock that is held by non-affiliates exceeds $700 million as of the last business day of our most recently completed second fiscal quarter, or (iii) the date on which we have issued more than $1 billion in non-convertible debt during the preceding three year period. An independent assessment of the effectiveness of our internal control over financial reporting could detect problems that our management’s assessment might not. Undetected material weaknesses in our internal control over financial reporting could lead to financial statement restatements and require us to incur the expense of remediation.
Moreover, we do not expect that disclosure controls or internal control over financial reporting will prevent all error and all fraud. A control system, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that the control system’s objectives will be met. Further, the design of a control system must reflect the fact that there are resource constraints and the benefits of controls must be considered relative to their costs. Because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all control issues and instances of fraud, if any, have been detected. Failure of our control systems to prevent error or fraud could materially adversely impact us.
Failure to build our finance infrastructure and improve our accounting systems and controls could impair our ability to comply with the financial reporting and internal control requirements for publicly traded companies.
As a public company, we operate in an increasingly demanding regulatory environment, which requires us to comply with applicable provisions of the Sarbanes-Oxley Act, and the related rules and regulations of the SEC, expanded disclosure requirements, accelerated reporting requirements and more complex accounting rules. Company responsibilities required by the Sarbanes-Oxley Act include establishing corporate oversight and adequate internal control over financial reporting and disclosure controls and procedures. Effective internal controls are necessary for us to produce reliable financial reports and are important to help prevent financial fraud.
We rely on consultants to perform certain of our accounting and financial reporting functions. We will need to hire additional finance personnel and build our financial infrastructure as we comply with public company reporting requirements, including complying with the applicable requirements of Section 404 of the Sarbanes-Oxley Act. We may be unable to do so on a timely basis.
| S-53 |
Upon dissolution of our company, you may not recoup all or any portion of your investment.
In the event of a liquidation, dissolution or winding-up of our company, whether voluntary or involuntary, the proceeds and/or assets of our company remaining after giving effect to such transaction, and the payment of all of our debts and liabilities will be distributed first to the holders of our Series A Preferred Stock and thereafter to the stockholders of common stock (including the holders of our Series A Preferred Stock on an “as converted” basis) on a pro rata basis. There can be no assurance that we will have available assets to pay to the holders of common stock, or any amounts, upon such a liquidation, dissolution or winding-up of our Company. In this event, you could lose some or all of your investment.
Our certificate of incorporation allows for our board to create new series of preferred stock without further approval by our stockholders, which could adversely affect the rights of the holders of our common stock.
Our board of directors has the authority to fix and determine the relative rights and preferences of preferred stock. We anticipate that our board of directors will have the authority to issue up to 8,392,000 additional shares of our preferred stock without further stockholder approval. As a result, our board of directors could authorize the issuance of a series of preferred stock that would grant to holders the preferred right to our assets upon liquidation, the right to receive dividend payments before dividends are distributed to the holders of common stock and the right to the redemption of the shares, together with a premium, prior to the redemption of our common stock. In addition, our board of directors could authorize the issuance of a series of preferred stock that has greater voting power than our common stock or that is convertible into our common stock, which could decrease the relative voting power of our common stock or result in dilution to our existing stockholders.
Anti-takeover provisions of our certificate of incorporation, our bylaws and Delaware law could make an acquisition of us, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove the current members of our board and management.
Certain provisions of our amended and restated certificate of incorporation and bylaws could discourage, delay or prevent a merger, acquisition or other change of control that stockholders may consider favorable, including transactions in which you might otherwise receive a premium for your Shares. Furthermore, these provisions could prevent or frustrate attempts by our stockholders to replace or remove members of our board of directors. These provisions also could limit the price that investors might be willing to pay in the future for our common stock, thereby depressing the market price of our common stock. Stockholders who wish to participate in these transactions may not have the opportunity to do so. These provisions, among other things:
| ● | they provide that special meetings of stockholders may be called only by the board of directors, President or our Chairman of the Board of Directors, or at the request in writing by stockholders of record owning at least fifty (50%) percent of the issued and outstanding voting shares of common stock; | |
| ● | they do not include a provision for cumulative voting in the election of directors. Under cumulative voting, a minority stockholder holding a sufficient number of shares may be able to ensure the election of one or more directors. The absence of cumulative voting may have the effect of limiting the ability of minority stockholders to effect changes in our board of directors; and | |
| ● | they allow us to issue, without stockholder approval, up to 10,000,000 shares of preferred stock (of which up to 8,392,000 shares remain available for issuance) that could adversely affect the rights and powers of the holders of our common stock. |
In addition, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, or DGCL, which may, unless certain criteria are met, prohibit large stockholders, in particular those owning 15% or more of the voting rights on our common stock, from merging or combining with us for a prescribed period of time.
| S-54 |
Our ability to use our net operating loss carryforwards and certain other tax attributes may be limited.
As a result of our merger with Aquarius Biotechnologies, Inc., our ability to utilize our U.S. federal net operating loss, carryforwards and U.S. federal tax credits may be limited under Sections 382 of the Internal Revenue Code of 1986, as amended. The limitations apply if an “ownership change,” as defined by Section 382 and Section 383, occurs. Generally, an ownership change occurs if the percentage of the value of the stock that is owned by one or more direct or indirect “five percent shareholders” increases by more than 50 percentage points over their lowest ownership percentage at any time during the applicable testing period (typically three years). In addition, future changes in our stock ownership, which may be outside of our control, may trigger an “ownership change” and, consequently, Section 382 and Section 383 limitations. As a result, if we earn net taxable income, our ability to use our pre-change net operating loss carryforwards and other tax attributes to offset U.S. federal taxable income may be subject to limitations, which could potentially result in increased future tax liability to us. The Tax Act, among other things, imposes significant additional limitations on the deductibility of interest and limits net operating loss (NOL) deductions.
Risks Related to this Offering
Management will have broad discretion as to the use of the proceeds from this offering, and we may not use the proceeds effectively.
Because we have not designated the amount of net proceeds received by us from this offering to be used for any particular purpose, our management will have broad discretion as to the application of the net proceeds from this offering and could use them for purposes other than those contemplated at the time of the offering. Our management may use the net proceeds for corporate purposes that may not improve our financial condition or market value.
You will experience immediate and substantial dilution as a result of this offering and may experience additional dilution in the future.
You will incur immediate and substantial dilution as a result of this offering. After giving effect to the sale by us of shares offered in this offering at an assumed public offering price of $1.10 per share of common stock, and after deducting underwriting discounts and commissions and expenses and other estimated offering expenses payable by us, investors in this offering can expect an immediate dilution of approximately $0.83 per share. See “Dilution” below for a more detailed discussion of the dilution you will incur if you purchase our common stock in the offering.
You may experience future dilution as a result of future equity offerings.
To raise additional capital, we may in the future offer additional shares of our common stock or other securities convertible into or exchangeable for our common stock at prices that may not be the same as the price per share in this offering. We may sell shares or other securities in any other offering at a price per share that is less than the price per share paid by investors in this offering, and investors purchasing shares or other securities in the future could have rights superior to existing stockholders. The price per share at which we sell additional shares of our common stock, or securities convertible or exchangeable into common stock, in future transactions may be higher or lower than the price per share paid by investors in this offering (on a fully-converted basis). Furthermore, sales of a substantial number of shares of our common stock in the public markets, or the perception that such sales could occur, could depress the market price of our common stock.
You may be subject to substantial dilution by exercises of outstanding options and warrants, conversion of preferred shares and by the future issuance of common stock to the former stockholders of Aquarius pursuant to the terms of the merger agreement.
As of March 4, 2019, we had outstanding options to purchase an aggregate of 16,251,796 shares of our common stock at a weighted average exercise price of $1.12 per share and warrants to purchase an aggregate of 5,802,256 shares of our common stock at a weighted average exercise price of $0.61 per share. In addition, as of March 4, 2019, we had 1,467,858 shares of Series A Preferred Stock outstanding and 4,819 shares of Series B Preferred Stock outstanding. Each share of Series A Preferred Stock may be converted into 10 shares of common stock upon the request of the holder and each share of Series B Preferred Stock may be converted into 2,000 shares of common stock upon the request of the holder. In addition, pursuant to the terms of the merger agreement with Aquarius Biotechnologies, Inc., we will be required to issue up to an additional 3,000,000 shares of our common stock upon the achievement of certain milestones. The milestone consideration consists of (i) 1,500,000 shares issuable upon the dosing of the first patient in a Phase 3 trial sponsored by us for a product utilizing the lipid nano-crystal (LNC) delivery technology platform and (ii) 1,500,000 shares issuable upon FDA approval of the first NDA submitted by us for a product utilizing the lipid nano-crystal (LNC) delivery technology platform. The issuance of additional shares of common stock upon the conversion of outstanding Series A Preferred Stock, Series B Preferred Stock or exercise of outstanding options and warrants or pursuant to the terms of the merger agreement will result in further dilution of the value of our shares.
| S-55 |
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus supplement, the accompanying prospectus and the documents incorporated by reference herein contain forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Forward-looking statements represent our management’s judgment regarding future events. In many cases, you can identify forward- looking statements by terminology such as “may,” “will,” “should,” “plan,” “expect,” “anticipate,” “estimate,” “predict,” “intend,” “potential” or “continue” or the negative of these terms or other words of similar import, although some forward-looking statements are expressed differently. All statements other than statements of historical fact included in this prospectus supplement, the accompanying prospectus and the documents incorporated by reference herein regarding our financial position, business strategy and plans or objectives for future operations are forward-looking statements. Without limiting the broader description of forward-looking statements above, we specifically note that statements regarding potential drug candidates, their potential therapeutic effect, the possibility of obtaining regulatory approval, our expected timing for completing clinical trials and clinical trial milestones for our drug candidates, our ability or the ability of our collaborators to manufacture and sell any products, market acceptance or our ability to earn a profit from sales or licenses of any drug candidate or to discover new drugs in the future are all forward-looking in nature. We cannot guarantee the accuracy of forward-looking statements, and you should be aware that results and events could differ materially and adversely from those described in the forward-looking statements due to a number of factors, including, but not limited to:
| ● | our ability to raise additional capital to fund our operations and to develop our product candidates; |
| ● | our anticipated timing for preclinical development, regulatory submissions, commencement and completion of clinical trials and product approvals; |
| ● | our history of operating losses in each year since inception and the expectation that we will continue to incur operating losses for the foreseeable future; |
| ● | our dependence on product candidates, including MAT9001, which are still in an early development stage; |
| ● | our reliance on proprietary LNC drug delivery technology platform, which is licensed to us by Rutgers University; |
| ● | our ability to manufacture GMP batches of our product candidates, including MAT9001 and MAT2203, which are required for preclinical and clinical trials and, subsequently, if regulatory approval is obtained for any of our products, our ability to manufacture commercial quantities; |
| ● | our ability to complete required clinical trials for our lead product candidate and other product candidates and obtain approval from the FDA or other regulatory agents in different jurisdictions; |
| ● | our expectations of the attributes of our product and development candidates, including pharmaceutical properties, efficacy, safety and dosing regimens including our anticipated market advantages and product differentiation of MAT9001, and its potential to become a best-in-class omega-3 therapeutic for the treatment of severe hypertriglyceridemia and potential to expand MAT9001’s indication to the treatment of high TGs (200-499 mg/dL); |
| ● | our dependence on third-parties, including third-parties to manufacture products and third-party contract research organizations (including, without limitation, the National Institutes of Health (NIH)) to conduct our clinical trials; |
| ● | our ability to maintain or protect the validity of our patents and other intellectual property; |
| ● | our ability to retain and recruit key personnel; |
| ● | our ability to internally develop new inventions and intellectual property; |
| ● | interpretations of current laws and the passages of future laws; |
| S-56 |
| ● | our lack of a sales and marketing organization and our ability to commercialize products, if we obtain regulatory approval, whether alone or through potential future collaborators; |
| ● | our ability to successfully commercialize, and our expectations regarding future therapeutic and commercial potential with respect to, our product candidates; |
| ● | the accuracy of our estimates regarding expenses, ongoing losses, future revenue, capital requirements and our needs for or ability to obtain additional financing; |
| ● | developments and projections relating to our competitors or our industry; and |
| ● | our ability to adequately support growth. |
You should also consider carefully the statements set forth in the section entitled “Risk Factors” in this prospectus supplement and in our Annual Report on Form 10-K for the year ended December 31, 2017, respectively, as updated by any other document that we subsequently filed with the Securities and Exchange Commission and that is incorporated by reference into this prospectus supplement, which address various factors that could cause results or events to differ from those described in the forward-looking statements. All subsequent written and oral forward-looking statements attributable to us or to persons acting on our behalf are expressly qualified in their entirety by the applicable cautionary statements. We have no plans to update these forward-looking statements.
| S-57 |
We estimate that the net proceeds received by us from this offering, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us, will be approximately $27.8 million. If the underwriters exercise their option to purchase additional shares in full, we estimate that the net proceeds from this offering will be approximately $32.0 million, after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us.
We currently intend to use the net proceeds from this offering primarily for ongoing development activities for our product candidates, namely MAT9001, and our LNC platform delivery technology and for working capital and other general corporate purposes.
This expected use of the net proceeds from this offering represents our intentions based upon our current plans and business conditions, and our management will retain broad discretion as to the ultimate allocation of the proceeds. Pending the use of the net proceeds of this offering, we intend to invest the funds in short-term, investment grade, interest-bearing securities.
| S-58 |
Purchasers of common stock in this offering will experience immediate dilution to the extent of the difference between the public offering price per share of common stock and the net tangible book value per share of common stock immediately after this offering.
Our net tangible book value as of September 30, 2018 was approximately $5.9 million, or $0.06 per share of common stock. Net tangible book value per share after this offering gives effect to the sale by us in this offering of 27,272,727 shares of common stock, at the assumed public offering price of $1.10 per share, after deducting underwriting discounts and commissions and estimated expenses payable by us. Our net tangible book value as of September 30, 2018, after giving effect to this offering as described above, would have been approximately $33.7 million, or $0.27 per share of common stock. This represents an immediate increase in pro forma net tangible book value of $0.21 per share to existing stockholders and an immediate dilution of $0.83 per share to new investors purchasing our common stock in this offering. The following table illustrates the per share dilution:
| Assumed public offering price per share | $ | 1.10 | ||||||
| Net tangible book value per share as of September 30, 2018 | $ | 0.06 | ||||||
| Increase in net tangible book value per share attributable to this offering | $ | 0.21 | ||||||
| Pro forma net tangible book value per share as of September 30, 2018, after giving effect to this offering | $ | 0.27 | ||||||
| Dilution per share to new investors in this offering | $ | 0.83 |
The above table does not include the following, as of September 30, 2018:
| ● | 11,455,029 shares issuable upon the exercise of outstanding stock options with a weighted-average exercise price of $1.21 per share; | |
| ● | 14,678,580 shares issuable upon conversion of outstanding shares of Series A Preferred Stock; | |
| ● | 2,348,573 shares issuable upon payment of dividends on Series A Preferred Stock; | |
| ● | 14,006,000 shares issuable upon conversion of outstanding shares of Series B Preferred Stock; | |
| ● | 5,802,256 shares issuable upon the exercise of outstanding warrants with a weighted-average exercise price of $0.61 per share; | |
| ● | 4,890,183 shares available for future issuance under our equity compensation plans; and | |
| ● | 3,000,000 shares issuable upon achievement of certain milestones pursuant to the terms of our merger agreement with Aquarius Biotechnologies, Inc. |
To the extent that outstanding options or warrants are exercised or we issue shares pursuant to the terms of our merger agreement, new equity awards are issued under our 2013 Equity Incentive Plan, or we issue additional shares of common stock in the future, there may be further dilution to investors participating in this offering. In addition, we need to raise additional capital. If we raise additional capital through the sale of equity or convertible debt securities, the issuance of these securities could result in further dilution to our stockholders.
| S-59 |
Subject to the terms and conditions set forth in the underwriting agreement between us and the underwriters named below, we have agreed to sell to each underwriter, and each underwriter has agreed to purchase from us, the shares of common stock shown opposite its name below:
| Underwriter | Number of Shares | |||
| BTIG, LLC | 27,272,727 | |||
Commission and Expenses
The underwriters have agreed to purchase the shares of common stock from us at a price of $1.034 per share, which will result in approximately $28.2 million of proceeds to us before expenses. The underwriters propose to offer the shares of common stock from time to time for sale in one or more transactions on NYSE American, in the over-the-counter market, through negotiated transactions or otherwise, at market prices prevailing at the time of sale, at prices related to prevailing market prices or at negotiated prices, subject to receipt and acceptance by them and subject to their right to reject any order in whole or in part. The difference between the price at which the underwriters purchase shares and the price at which the underwriters resell such shares may be deemed underwriting compensation. The underwriters may effect such transactions by selling shares of common stock to or through dealers, and as such dealers may receive compensation in the form of discounts, concessions or commission from the underwriters and/or purchases of shares of common stock for whom they may act as agents or to who they may sell as principal. We have agreed to reimburse the underwriters up to $15,000 in connection with their expenses related to the clearance of this offering with the Financial Industry Regulatory Authority.
We estimate expenses payable by us in connection with this offering will be approximately $0.4 million.
Listing
Our common stock is listed on the NYSE American LLC under the trading symbol “MTNB”.
No Sales of Similar Securities
We, our officers and our directors have agreed, subject to certain specified exceptions, not to directly or indirectly, for a period of 90 days after the date of the underwriting agreement:
| ● | sell, offer, contract or grant any option to sell (including any short sale), pledge, transfer, establish an open “put equivalent position” within the meaning of Rule 16a-l(h) under the Exchange Act, or |
| ● | otherwise dispose of any shares of common stock, options or warrants to acquire shares of common stock, or securities exchangeable or exercisable for or convertible into shares of common stock currently or hereafter owned either of record or beneficially, or |
| ● | publicly announce an intention to do any of the foregoing for a period of 90 days after the date of this prospectus supplement without the prior written consent of BTIG, LLC. |
In addition, we and each such person agrees that, without the prior written consent of BTIG, LLC, we or such other person will not, during the restricted period, make any demand for, or exercise any right with respect to, the registration of any shares of common stock or any security convertible into or exercisable or exchangeable for common stock.
| S-60 |
The restrictions in the immediately preceding paragraph do not apply in certain circumstances, including, among others:
| ● | the sale of common stock in this offering, | |
|
● | in connection with employee benefit plans, equity compensation plans or other compensation plans as in existence on the date of this prospectus and as described in the prospectus, |
| ● | in connection with currently outstanding options, warrants or rights, |
| ● | the consummation by us of a strategic partnership, joint venture, collaboration or acquisition or license of any business products or technology, and |
| ● | the filing by us of any registration statement on Form S-8 in respect of any equity compensation plans or arrangements maintained by us. |
BTIG, LLC may, in its sole discretion and at any time or from time to time before the termination of the 90-day period, release all or any portion of the securities subject to lock-up agreements. There are no existing agreements between the underwriters and any of our shareholders who will execute a lock-up agreement, providing consent to the sale of shares prior to the expiration of the lock-up period.
Market Making, Stabilization and Other Transactions
The underwriters may make a market in the common stock as permitted by applicable laws and regulations. However, the underwriters are not obligated to do so, and each underwriter may discontinue any market-making activities at any time without notice in its sole discretion. Accordingly, no assurance can be given as to the liquidity of the trading market for the common stock, that you will be able to sell any of the common stock held by you at a particular time or that the prices that you receive when you sell will be favorable.
The underwriters have advised us that they, pursuant to Regulation M under the Exchange Act, and certain persons participating in this offering, may engage in short sale transactions, stabilizing transactions, syndicate covering transactions or the imposition of penalty bids in connection with this offering. These activities may have the effect of stabilizing or maintaining the market price of our common stock at a level above that which might otherwise prevail in the open market. Establishing short sales positions may involve either “covered” short sales or “naked” short sales.
“Covered” short sales are sales made in an amount not greater than the underwriters’ option to purchase additional shares of our common stock in this offering. The underwriters may close out any covered short position by either exercising their option to purchase additional shares of our common stock or purchasing shares of our common stock in the open market. In determining the source of shares to close out the covered short position, the underwriters will consider, among other things, the price of shares of common stock available for purchase in the open market as compared to the price at which they may purchase shares through the option to purchase additional shares.
“Naked” short sales are sales in excess of the option to purchase additional shares of our common stock. The underwriters must close out any naked short position by purchasing shares in the open market. A naked short position is more likely to be created if the underwriters are concerned that there may be downward pressure on the price of the shares of our common stock in the open market after pricing that could adversely affect investors who purchase in this offering.
A stabilizing bid is a bid for the purchase of shares of common stock on behalf of the underwriters for the purpose of fixing or maintaining the price of the common stock. A syndicate covering transaction is the bid for or the purchase of shares of common stock on behalf of the underwriters to reduce a short position incurred by the underwriters in connection with this offering. Similar to other purchase transactions, the underwriters’ purchases to cover the syndicate short sales may have the effect of raising or maintaining the market price of our common stock or preventing or retarding a decline in the market price of our common stock. As a result, the price of our common stock may be higher than the price that might otherwise exist in the open market. A penalty bid is an arrangement permitting the underwriters to reclaim the selling concession otherwise accruing to a syndicate member in connection with this offering if the common stock originally sold by such syndicate member are purchased in a syndicate covering transaction and therefore have not been effectively placed by such syndicate member.
| S-61 |
Neither we, nor the underwriters, make any representation or prediction as to the direction or magnitude of any effect that the transactions described above may have on the price of our common stock. The underwriters are not obligated to engage in these activities and, if commenced, may end any of these activities at any time.
Passive Market Making
The underwriters may also engage in passive market-making transactions in our common stock on NYSE American in accordance with Rule 103 of Regulation M during a period before the commencement of offers or sales of shares of our common stock in this offering and extending through the completion of distribution. A passive market maker must display its bid at a price not in excess of the highest independent bid of that security. However, if all independent bids are lowered below the passive market maker’s bid, that bid must then be lowered when specified purchase limits are exceeded. Passive market making may cause the price of our common stock to be higher than the price that otherwise would exist in the open market in the absence of those transactions. The underwriters are not required to engage in passive market making and, if commenced, may end passive market making activities at any time.
Electronic Distribution
A prospectus in electronic format may be made available by e-mail or on the web sites or through online services maintained by the underwriters, selling group members (if any) or their affiliates. The underwriters may agree with us to allocate a specific number of shares of common stock for sale to online brokerage account holders. Any such allocation for online distributions will be made by the underwriters on the same basis as other allocations. Other than the prospectus in electronic format, the information on the underwriters’ web sites and any information contained in any other web site maintained by any of the underwriters is not part of this prospectus supplement or the accompanying prospectus, has not been approved and/or endorsed by us or the underwriters and should not be relied upon by investors.
Other Activities and Relationships
The underwriters and certain of their respective affiliates are full service financial institutions engaged in a wide range of activities for their own accounts and the accounts of customers, which may include, among other things, corporate finance, mergers and acquisitions, merchant banking, equity and fixed income sales, trading and research, derivatives, foreign exchange, futures, asset management, custody, clearance and securities lending. The underwriters and certain of their affiliates have, from time to time, performed, and may in the future perform, various investment banking and financial advisory services for us and our affiliates, for which they received or will receive customary fees and expenses.
In addition, in the ordinary course of their various business activities, the underwriters and certain of their respective affiliates may, directly or indirectly, hold long or short positions, trade and otherwise conduct such activities in or with respect to debt or equity securities and/or bank debt of, and/or derivative products. Such investment and securities activities may involve our securities and instruments. The underwriters and certain of their respective affiliates may also make investment recommendations or publish or express independent research views in respect of such securities or instruments and may at any time hold, or recommend to clients that they acquire, long or short positions in such securities and instruments.
Stamp Taxes
If you purchase shares of common stock offered in this prospectus supplement, you may be required to pay stamp taxes and other charges under the laws and practices of the country of purchase, in addition to the offering price listed on the cover page of this prospectus supplement.
| S-62 |
Lowenstein Sandler LLP, New York, New York, will provide us with an opinion as to the validity of the shares of common stock offered by this prospectus supplement and the accompanying prospectus. Partners of the firm beneficially own an aggregate of 145,000 shares of common stock, 5,000 shares of Series A Preferred Stock, and warrants to purchase 20,000 shares of our common stock with an exercise price of $0.75 per share. Goodwin Procter LLP, New York, New York has acted as counsel for the underwriters in connection with certain legal matters related to this offering.
The consolidated balance sheets of Matinas BioPharma Holdings, Inc. and Subsidiaries as of December 31, 2017 and 2016, and the related consolidated statements of operations, stockholders’ equity, and cash flows for each of the years in the three-year period ended December 31, 2017, have been audited by EisnerAmper LLP, independent registered public accounting firm, as stated in their report which is incorporated herein by reference. Such financial statements have been incorporated herein by reference in reliance on the report of such firm given upon their authority as experts in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
We file annual, quarterly and current reports, proxy statements and other information with the SEC. We have also filed a registration statement on Form S-3, including exhibits, under the Securities Act with respect to the securities offered by this prospectus supplement and the accompanying prospectus. This prospectus supplement and the accompanying prospectus are a part of the registration statement but do not contain all of the information included in the registration statement or the exhibits. You can also find our public filings with the SEC on the Internet at a web site maintained by the SEC located at http://www.sec.gov.
| S-63 |
INCORPORATION OF CERTAIN INFORMATION BY REFERENCE
The SEC allows us to incorporate by reference the information and reports we file with it, which means that we can disclose important information to you by referring you to these documents. The information incorporated by reference is an important part of this prospectus supplement, and information that we file later with the SEC will automatically update and supersede the information already incorporated by reference. We are incorporating by reference the documents listed below, which we have already filed with the SEC, and any future filings we make with the SEC under Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act, except as to any portion of any future report or document that is not deemed filed under such provisions, prior to the completion or termination of this offering:
| ● | our Annual Report on Form 10-K for the year ended December 31, 2017 filed with the SEC on March 16, 2018, as amended by our Form 10-K/A, filed with the SEC on May 25, 2018 and our Form 10-K/A, filed with the SEC on June 14, 2018; | |
| ● | our Quarterly Reports on Form 10-Q for the periods ended March 31, 2018, June 30, 2018 and September 30, 2018 filed with the SEC on May 9, 2018, August 9, 2018 and November 9, 2018, respectively; | |
| ● | our Current Reports on Form 8-K filed with the SEC on January 8, 2018, March 27, 2018, May 25, 2018, June 18, 2018, June 19, 2018, September 6, 2018, October 15, 2018, November 2, 2018, January 3, 2019 and February 25, 2019 (other than information “furnished” under Items 2.02 or 7.01 (or corresponding information furnished under Item 9.01 or included as an exhibit); and | |
| ● | the description of our common stock contained in the Registration Statement on Form 8-A, declared effective on August 12, 2013 (including any amendment or report filed with the SEC for the purpose of updating such description). |
Upon request, we will provide, without charge, to each person, including any beneficial owner, to whom a copy of this prospectus is delivered a copy of the documents incorporated by reference into this prospectus supplement. You may request a copy of these filings, and any exhibits we have specifically incorporated by reference as an exhibit in this prospectus supplement, at no cost by writing or telephoning us at the following address:
Investor Relations Department
Matinas BioPharma Holdings, Inc.
1545 Route 206 South
Suite 302
Bedminster, NJ 07921
Telephone number: 908-443-1860
Any statement contained in this prospectus supplement or in a document incorporated or deemed to be incorporated by reference into this prospectus supplement will be deemed to be modified or superseded for purposes of this prospectus supplement to the extent that a statement contained in this prospectus supplement or any other subsequently filed document that is deemed to be incorporated by reference into this prospectus supplement modifies or supersedes the statement. Any statement so modified or superseded will not be deemed, except as so modified or superseded, to constitute a part of this prospectus supplement.
You should rely only on information contained in, or incorporated by reference into, this prospectus supplement and the accompanying prospectus. We have not authorized anyone to provide you with information different from that contained in this prospectus supplement and the accompanying prospectus or incorporated by reference in this prospectus supplement and the accompanying prospectus. We are not making offers to sell the securities in any jurisdiction in which such an offer or solicitation is not authorized or in which the person making such offer or solicitation is not qualified to do so or to anyone to whom it is unlawful to make such offer or solicitation..
| S-64 |
Prospectus

$150,000,000
Common Stock
Preferred Stock
Warrants
Debt Securities
Subscription Rights
Units
We may offer, issue and sell from time to time together or separately, in one or more offerings, any combination of (i) our common stock, (ii) our preferred stock, which we may issue in one or more series, (iii) warrants, (iv) senior or subordinated debt securities, (v) subscription rights and (vi) units, consisting of any combination of the securities listed above. We will describe in a prospectus supplement the securities we are offering and selling, as well as the specific terms of the securities.
The aggregate public offering price of the securities that we are offering will not exceed $150,000,000. We will offer the securities in an amount and on terms that market conditions will determine at the time of the offering. Our common stock is listed on the NSYE MKT under the symbol “MTNB.” The last reported sale price for our common stock on March 30, 2017 was $2.75 per share. You are urged to obtain current market quotations of our common stock. We have no preferred stock, warrants, debt securities, subscription rights or units listed on any market. Each prospectus supplement will indicate if the securities offered thereby will be listed on any securities exchange.
Investing in our securities involves risk. You should carefully consider the risks that we have described under the section captioned “Risk Factors” in this prospectus on page 3 before buying our Securities.
Should we offer any of the securities described in this prospectus, we will provide you with the specific terms of the particular securities being offered in supplements to this prospectus. You should read this prospectus and any supplement, together with additional information described under the headings “Additional Information” and “Incorporation of Certain Information by Reference,” carefully before you invest. This prospectus may not be used to sell securities unless accompanied by a prospectus supplement.
We may sell these securities directly to our stockholders or to purchasers or through agents on our behalf or through underwriters or dealers as designated from time to time. If any agents or underwriters are involved in the sale of any of these securities, the applicable prospectus supplement will provide the names of the agents or underwriters and any applicable fees, commissions or discounts.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful and complete. Any representation to the contrary is a criminal offense.
The date of this prospectus is April 12, 2017
Unless the context indicates otherwise, “Matinas”, “the Company”, “we”, “us”, and “our” as used herein refers to Matinas BioPharma Holdings, Inc. and its wholly owned subsidiaries, Matinas BioPharma Inc. and Matinas BioPharma Nanotechnologies, Inc.
You may only rely on the information contained in this prospectus or that we have referred you to. We have not authorized anyone to provide you with different information. This prospectus does not constitute an offer to sell or a solicitation of an offer to buy any securities other than the securities offered by this prospectus. This prospectus and any future prospectus supplement do not constitute an offer to sell or a solicitation of an offer to buy any securities in any circumstances in which such offer or solicitation is unlawful. Neither the delivery of this prospectus or any prospectus supplement nor any sale made hereunder shall, under any circumstances, create any implication that there has been no change in our affairs since the date of this prospectus or such prospectus supplement or that the information contained by reference to this prospectus or any prospectus supplement is correct as of any time after its date.
This prospectus is part of a registration statement that we filed with the Securities and Exchange Commission (“SEC”) using a “shelf” registration process. Under this shelf registration process, we may from time to time offer and sell, in one or more offerings, any or all of the securities described in this prospectus, separately or together, up to an aggregate initial offering price of $150,000,000. This prospectus provides you with a general description of our securities being offered. When we issue the securities being offered by this prospectus, we will provide a prospectus supplement that will contain specific information about the terms of that offering. The prospectus supplement may also add, update or change information contained in this prospectus. You should read both this prospectus and any prospectus supplement together with additional information described under the heading “Additional Information” and “Incorporation of Certain Information by Reference.”
The following summary highlights some information from this prospectus. It is not complete and does not contain all of the information that you should consider before making an investment decision. You should read this entire prospectus, including the “Risk Factors” section on page 3, the financial statements and related notes and the other more detailed information appearing elsewhere or incorporated by reference into this prospectus.
We currently do not own or license any U.S. federal trademark registrations or applications. Some trademarks referred to in this prospectus are referred to without the ® and ™ symbols, but such references should not be construed as any indicator that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto.
About Us
We are a clinical-stage biopharmaceutical company focused on developing innovative anti-infectives for orphan indications. Our product and development candidates are derived using our unique and proprietary lipid-crystal nano-particle, or cochleate, formulation platform delivery technology. Our proprietary cochleate delivery technology platform, licensed from Rutgers University on an exclusive worldwide basis, nano-encapsulates drugs and is designed to make these drugs orally bioavailable, well tolerated and safer and less toxic while providing targeted and safe delivery of pharmaceuticals directly to the site of infection or inflammation. We believe our cochleate technology provides us with an efficient and broadly applicable drug delivery platform, with particular utility in diseases and conditions in which the immune system plays a significant modulation role and where the immune system facilitates the active transport of our lipid crystal nano-particles throughout the body.
Currently, we are focused on the anti-infective market and on drug candidates which we believe demonstrate the value and innovation associated with our unique cochleate delivery platform technology while potentially providing significant health economic benefit to the health care system. We believe initially focusing on the anti-infective market has distinct advantages for the development of products which meet significant unmet medical need, including:
| ● | a current regulatory environment which provides small development and clinical stage companies incentives such as significant periods of regulatory marketing exclusivity and opportunities to reduce development cost and timeline to market for anti-infective drug candidates; |
| ● | traditional high correlation between efficacy and safety data in preclinical animal models and the outcome of human clinical trials with anti-infective product candidates; |
| ● | attractive commercial opportunities for anti-infective product differentiated in safety profile, mode of action and oral bioavailability positioned against current therapies with significant side effects, or drug to drug interactions, limited efficacy and intravenous delivery resulting in lack of convenience, compliance and at a significant burden to the cost of healthcare; and |
| ● | an ability to commercialize anti-infective products with a focused and cost-efficient sales and marketing organization. |
| 1 |
MAT2203 and MAT2501
We leveraged our platform cochleate delivery technology to develop two clinical-stage products that we believe have the potential to become best-in-class drugs. Our lead product candidate MAT2203 is an orally-administered cochleate formulation of a broad spectrum anti-fungal drug called amphotericin B. We are initially developing MAT2203 for the treatment of serious fungal infections as well as the prevention of invasive fungal infections (IFIs) due to immunosuppressive therapy. We are currently conducting two Phase 2 clinical trials involving MAT2203 and expect to report interim results from our open label NIH run Phase 2a clinical trial and topline results from our ongoing Phase 2 study of MAT2203 in Vulvovaginal Candidiasis in the first half of 2017.
Our second clinical stage product candidate is MAT2501, an orally administered, encochleated formulation of the broad spectrum aminoglycoside antibiotic amikacin which may be used to treat different types of multidrug-resistant bacteria, including non-tuberculous mycobacterium infections (NTM), as well as various multidrug-resistant gram negative and intracellular bacterial infections. We recently completed and announced topline results from a Phase 1 single escalating dose clinical trial of MAT2501 in healthy volunteers in which no serious adverse events were reported and where oral administration of MAT2501 at all tested doses yielded blood levels that were well below the safety levels recommended for injected amikacin, supporting further development of MAT2501 for the treatment of NTM infections.
Implications of Being an Emerging Growth Company
We are an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act of 2012. We will remain an emerging growth company until December 31, 2019, or until the earliest of (i) the last day of the first fiscal year in which our annual gross revenues exceed $1 billion, (ii) the date that we become a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act, which would occur if the market value of our common stock that is held by non-affiliates exceeds $700 million as of the last business day of our most recently completed second fiscal quarter, or (iii) the date on which we have issued more than $1 billion in non-convertible debt during the preceding three year period.
For
as long as we remain an “emerging growth company,” we intend to take advantage of certain exemptions from various
reporting requirements that are applicable to public companies that are not “emerging growth companies” including,
but not limited to, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley
Act, reduced disclosure obligations regarding executive compensation and financial statements in our periodic reports and proxy
statements, and exemptions from the requirements of holding a nonbinding advisory vote to approve executive compensation and shareholder
approval of any golden parachute payments not previously approved. We will take advantage of these reporting exemptions until
we are no longer an “emerging growth company.”
Corporate Information
We were incorporated in Delaware under the name Matinas BioPharma Holdings, Inc. in May 2013. We have two operating subsidiaries: Matinas BioPharma, Inc., a Delaware corporation, and Matinas BioPharma Nanotechnologies, Inc., a Delaware corporation. Nereus BioPharma LLC, a Delaware limited liability company (and Matinas BioPharma’s predecessor) was formed on August 12, 2011. On February 29, 2012, Nereus BioPharma LLC converted from a limited liability company to a corporation and changed its name to Matinas BioPharma, Inc. In July 2013, Matinas BioPharma, Inc. entered into entered into a merger agreement (the “2013 Merger Agreement”) with Matinas Merger Sub, Inc., a Delaware corporation and our wholly owned subsidiary, or Merger Sub. Pursuant to the terms of the 2013 Merger Agreement, as a condition of and contemporaneously with the initial closing of the 2013 Private Placement, Merger Sub merged (the “2013 Merger”) with and into Matinas BioPharma and Matinas BioPharma became a wholly owned subsidiary of ours. After consummation of the Merger transaction, the management of Matinas BioPharma became the management of Holdings and the board representatives consisted of four former Board members of Matinas BioPharma and Mr. Adam Stern as the Aegis Capital Corp. nominee. Because Holdings was formed solely to effect the 2013 Merger and the 2013 Private Placement, with no operations, and assets consisting solely of cash and cash equivalents, we accounted for the 2013 Merger as a reverse acquisition. The legal acquirer Matinas BioPharma becomes the successor entity, and its historical results became the historical results for Holdings (the legal acquirer and the registrant). On January 29, 2015, we acquired Aquarius Biotechnologies Inc. which was subsequently renamed Matinas BioPharma Nanotechnologies, Inc.
Our principal executive offices are located at 1545 Route 206 South, Suite 302, Bedminster, New Jersey 07921, and our telephone number is (908) 443-1860. Our website address is www.matinasbiopharma.com. Our website and the information contained on, or that can be accessed through, our website will not be deemed to be incorporated by reference in, and are not considered part of, this prospectus. You should not rely on our website or any such information in making your decision whether to purchase our securities.
| 2 |
An investment in our securities involves a high degree of risk. Before making an investment decision, you should carefully consider the risks described under “Risk Factors” in the applicable prospectus supplement and in our most recent Annual Report on Form 10-K, and in our updates to those Risk Factors in our Quarterly Reports on Form 10-Q or Current Reports on Form 8-K following the most recent Form 10-K, and in all other information appearing in this prospectus or incorporated by reference into this prospectus and any applicable prospectus supplement. The material risks and uncertainties that management believes affect us will be described in those documents. In addition to those risk factors, there may be additional risks and uncertainties of which management is not aware or focused on or that management deems immaterial. Our business, financial condition or results of operations could be materially adversely affected by any of these risks. The trading price of our securities could decline due to any of these risks, and you may lose all or part of your investment. This prospectus is qualified in its entirety by these risk factors.
This prospectus, including the documents that we incorporate by reference, contains forward-looking statements as that term is defined in the federal securities laws. The events described in forward-looking statements contained in this prospectus, including the documents that we incorporate by reference, may not occur. Generally, these statements relate to our business plans or strategies, projected or anticipated benefits or other consequences of our plans or strategies, financing plans, projected or anticipated benefits from acquisitions that we may make, or projections involving anticipated revenues, earnings or other aspects of our operating results or financial position, and the outcome of any contingencies. Any such forward-looking statements are based on current expectations, estimates and projections of management. We intend for these forward-looking statements to be covered by the safe-harbor provisions for forward-looking statements. Words such as “may,” “expect,” “believe,” “anticipate,” “project,” “plan,” “intend,” “estimate,” and “continue,” and their opposites and similar expressions are intended to identify forward-looking statements. We caution you that these statements are not guarantees of future performance or events and are subject to a number of uncertainties, risks and other influences, many of which are beyond our control that may influence the accuracy of the statements and the projections upon which the statements are based. Factors that may affect our results include, but are not limited to, the risks and uncertainties discussed in the “Risk Factors” section on page 3 of this prospectus, in our most recent Annual Report on Form 10-K or in other reports we file with the Securities and Exchange Commission.
Any one or more of these uncertainties, risks and other influences could materially affect our results of operations and whether forward-looking statements made by us ultimately prove to be accurate. Our actual results, performance and achievements could differ materially from those expressed or implied in these forward-looking statements. We undertake no obligation to publicly update or revise any forward-looking statements, whether from new information, future events or otherwise.
You should rely only on the information in this prospectus. We have not authorized any other person to provide you with different information. If anyone provides you with different or inconsistent information, you should not rely upon it. You should assume that the information in this prospectus was accurate on the date of the front cover of this prospectus only. Our business, financial condition, results of operations and prospects may have changed since that date.
Unless we inform you otherwise in the prospectus supplement, we will use the net proceeds from the sale of the securities offered by this prospectus and the exercise price from the exercise of any convertible securities, if any, for working capital and general corporate purposes, which may include research and development funding, funding for clinical trials for our product candidates, manufacturing, acquisitions or investments in businesses, products or technologies that are complementary to our own and capital expenditures. Pending their uses, we intend to invest the net proceeds of this offering in interest-bearing bank accounts or in short-term, interest-bearing, investment-grade securities.
| 3 |
General
The descriptions of the securities contained in this prospectus, together with the applicable prospectus supplements, summarize all of the material terms and provisions of the various types of securities that we may offer. We will describe in the applicable prospectus supplement relating to any securities the particular terms of the securities offered by that prospectus supplement. If any particular terms of a security described in the applicable prospectus supplement differ from any of the terms described herein, then the terms described herein will be deemed superseded by the terms set forth in that prospectus supplement. We will also include in the prospectus supplement information, where applicable, about material United States federal income tax considerations relating to the securities, and the securities exchange, if any, on which the securities will be listed.
We may sell from time to time, in one or more offerings:
| ● | common stock; |
| ● | preferred stock; |
| ● | debt securities; |
| ● | warrants to purchase shares of common stock, preferred stock or other securities; |
| ● | subscription rights, and |
| ● | units consisting of any combination of the securities listed above. |
In this prospectus, we refer to the common stock, preferred stock, debt securities, warrants, subscription rights and units collectively as “securities.” The total dollar amount of all securities that we may sell will not exceed $150,000,000.
If we issue debt securities at a discount from their original stated principal amount, then, for purposes of calculating the total dollar amount of all securities issued under this prospectus, we will treat the initial offering price of the debt securities as the total original principal amount of the debt securities.
This prospectus may not be used to consummate a sale of securities unless it is accompanied by a prospectus supplement.
General
Our authorized capital stock consists of:
| ● | 250,000,000 shares of common stock, par value $0.0001 per share; and |
| ● | 10,000,000 shares of Preferred Stock, par value $0.0001 per share, of which as of the date of this prospectus 1,600,000 shares have been designated as Series A Preferred Stock. |
As of close of business on March 30, 2017, 89,333,282 shares of common stock were issued and outstanding and 1,600,000 shares of Series A Preferred Stock were issued and outstanding.
| 4 |
The additional shares of our authorized stock available for issuance might be issued at times and under circumstances so as to have a dilutive effect on earnings per share and on the equity ownership of the holders of our common stock. The ability of our board of directors to issue additional shares of stock could enhance the board’s ability to negotiate on behalf of the stockholders in a takeover situation but could also be used by the board to make a change-in-control more difficult, thereby denying stockholders the potential to sell their shares at a premium and entrenching current management. The following description is a summary of the material provisions of our capital stock. You should refer to our amended and restated certificate of incorporation and by-laws, both of which are on file with the SEC as exhibits to previous SEC filings, for additional information. The summary below is qualified by provisions of applicable law.
Common Stock
Voting. The holders of our common stock are entitled to one vote for each share held of record on all matters on which the holders are entitled to vote (or consent to).
Dividends. The holders of our common stock are entitled to receive, ratably, dividends only if, when and as declared by our Board of Directors out of funds legally available therefor and after provision is made for each class of capital stock having preference over the common stock (including the common stock).
Liquidation Rights. In the event of our liquidation, dissolution or winding-up, the holders of our common stock are entitled to share, ratably, in all assets remaining available for distribution after payment of all liabilities and after provision is made for each class of capital stock having preference over the common stock (including the common stock).
Conversion Rights. The holders of our common stock have no conversion rights.
Preemptive and Similar Rights. The holders of our common stock have no preemptive or similar rights.
Redemption/Put Rights. There are no redemption or sinking fund provisions applicable to the common stock. All of the outstanding shares of our common stock are fully-paid and nonassessable.
| 5 |
Transfer Agent and Registrar
The transfer agent and registrar for our common stock is VStock Transfer, LLC.
Preferred Stock
We are authorized to issue up to 10,000,000 shares of preferred stock, par value $0.0001 per share, (of which 1,600,000 shares have been designated as Series A Preferred Stock) with such designations, rights, and preferences as may be determined from time to time by our Board of Directors. Accordingly, our Board of Directors is empowered, without stockholder approval, to issue preferred stock with dividend, liquidation, conversion, voting, or other rights that could adversely affect the voting power or other rights of the holders of our common stock. The issuance of preferred stock could have the effect of restricting dividends on our common stock, diluting the voting power of our common stock, impairing the liquidation rights of our common stock, or delaying or preventing a change in control of our company, all without further action by our stockholders.
Our board of directors has the authority, within the limitations and restrictions prescribed by law and without stockholder approval, to provide by resolution for the issuance of shares of preferred stock, and to fix the rights, preferences, privileges and restrictions thereof, including dividend rights, conversion rights, voting rights, terms of redemption, liquidation preference and the number of shares constituting any series of the designation of such series, by delivering an appropriate certificate of amendment to our amended and restated certificate of incorporation to the Delaware Secretary of State pursuant to the Delaware General Corporation Law (the “DGCL”). The issuance of preferred stock could have the effect of decreasing the market price of the common stock, impeding or delaying a possible takeover and adversely affecting the voting and other rights of the holders of our common stock.
If we offer a specific series of preferred stock under this prospectus, we will describe the terms of the preferred stock in the prospectus supplement for such offering and will file a copy of the certificate establishing the terms of the preferred stock with the SEC. To the extent required, this description will include:
| ● | the title and stated value; |
| ● | the number of shares offered, the liquidation preference per share and the purchase price; |
| ● | the dividend rate(s), period(s) and/or payment date(s), or method(s) of calculation for such dividends; |
| ● | whether dividends will be cumulative or non-cumulative and, if cumulative, the date from which dividends will accumulate; |
| ● | the procedures for any auction and remarketing, if any; |
| ● | the provisions for a sinking fund, if any; |
| ● | the provisions for redemption, if applicable; |
| ● | any listing of the preferred stock on any securities exchange or market; |
| ● | whether the preferred stock will be convertible into our common stock, and, if applicable, the conversion price (or how it will be calculated) and conversion period; |
| ● | whether the preferred stock will be exchangeable into debt securities, and, if applicable, the exchange price (or how it will be calculated) and exchange period; |
| ● | voting rights, if any, of the preferred stock; |
| ● | a discussion of any material and/or special U.S. federal income tax considerations applicable to the preferred stock; |
| ● | the relative ranking and preferences of the preferred stock as to dividend rights and rights upon liquidation, dissolution or winding up of the affairs of Matinas; and |
| ● | any material limitations on issuance of any class or series of preferred stock ranking senior to or on a parity with the series of preferred stock as to dividend rights and rights upon liquidation, dissolution or winding up of Matinas. |
| 6 |
Transfer Agent and Registrar for Preferred Stock
The transfer agent and registrar for any series or class of preferred stock will be set forth in each applicable prospectus supplement.
Series A Preferred Stock
In connection with the 2016 Private Placement, our Board of Directors created out of the authorized and unissued shares of our preferred stock, a series of preferred stock comprised of 1,600,000 shares of Series A Preferred Stock. We have 1,600,000 shares of our Series A Preferred Stock outstanding as of March 30, 2017, which are convertible into 16,000,000 shares of common stock based on the current conversion price.
Rank. The Series A Preferred Stock ranks above all other classes of stock outstanding as of the date hereof with respect to dividend rights and liquidation preferences.
Dividends. Holders of Series A Preferred Stock are entitled to receive cumulative dividends at the rate per share of 8% per annum, payable in shares of our common stock, which annual dividend will accumulate until such time as the shares of Series A Preferred Stock are converted, at which time the accumulated dividend will be satisfied by delivery of shares of common stock (“PIK Shares”) at a price per share of common stock equal to the then conversion price. Each holder of shares of Series A Preferred Stock will be entitled to receive dividends equal, on an as-if-converted to shares of common stock basis, to and in the same form as dividends actually paid on shares of our common stock when, as, and if such dividends are paid on shares of our common stock.
Liquidation. Upon any dissolution, liquidation or winding up, whether voluntary or involuntary, holders of Series A Preferred Stock will be entitled to (i) first receive distributions out of our assets in an amount per share equal to $5.00 (the “Stated Value”) plus all accrued and unpaid dividends, whether capital or surplus before any distributions shall be made on any shares of common stock and (ii) second, on an as-converted basis alongside the common stock.
Conversion. Upon the earlier of (i) notice by us to the holders that we have elected to convert all outstanding shares of Series A Preferred Stock; provided however that in the event we elect to force automatic conversion pursuant to this clause (i), the conversion date for purposes of calculating the accrued dividend is deemed to be July 29, 2019, which is the third anniversary of the initial closing, (ii) July 29, 2019, (iii) the approval of the MAT2203 product candidate by the U.S. Food and Drug Administration or the European Medicines Agency (the “Regulatory Approval”) or (iv) the Regulatory Approval of MAT2501 product candidate, without any action on the part of the holder (each of the foregoing, a “Mandatory Conversion Date”), all of the outstanding shares of Series A Preferred Stock will automatically convert to common stock (a “Mandatory Conversion”). In addition, each share of Series A Preferred Stock shall be convertible, at any time and from time to time at the option of the holder thereof, into that number of shares of common stock determined by dividing the Stated Value of such Series A Preferred Stock by the conversion price. The conversion price currently is $0.50 per share of common stock and is subject to adjustment described below.
Stock Dividends and Stock Splits. If we pay a stock dividend or otherwise make a distribution payable in shares of common stock on shares of common stock or any other common stock equivalents, subdivide or combine outstanding common stock, or reclassify common stock, the conversion price will be adjusted by multiplying the then conversion price by a fraction, the numerator of which shall be the number of shares of common stock outstanding immediately before such event, and the denominator of which shall be the number of shares outstanding immediately after such event.
Fundamental Transaction. If we effect a fundamental transaction, then upon any subsequent conversion of Series A Preferred Stock, the holder thereof shall have the right to receive, for each share of common stock that would have been issuable upon such conversion immediately prior to the occurrence of such fundamental transaction, the number of shares of the successor’s or acquiring corporation’s common stock or of our common stock, if we are the surviving corporation, and any additional consideration receivable as a result of such fundamental transaction by a holder of the number of shares of common stock into which shares of our Series A Preferred Stock is convertible immediately prior to such fundamental transaction. A fundamental transaction means: (i) our merger or consolidation with or into another entity, (ii) any sale of all or substantially all of our assets in one transaction or a series of related transactions, or (iii) any reclassification of our common stock or any compulsory share exchange by which common stock is effectively converted into or exchanged for other securities, cash or property.
| 7 |
Voting Rights. Except as otherwise provided in the Certificate of Designation or required by law, shares of our Series A Preferred Stock have no class voting rights. The Certificate of Designations provides that each share of Series A Preferred Stock will entitle its holder to vote with the common stock on an as-if converted to shares of common stock basis. Notwithstanding certain protections in the Certificate of Designations, Delaware law also provides holders of preferred stock with certain rights. The holders of the outstanding shares of Series A Convertible Preferred Stock generally will be entitled to vote as a class upon a proposed amendment to our certificate of incorporation if the amendment would:
| ● | increase or decrease the aggregate number of authorized shares of our Series A Convertible Preferred Stock; |
| ● | increase or decrease the par value of the shares of our Series A Convertible Preferred Stock; or |
| ● | alter or change the powers, preferences, or special rights of the shares of our Series A Convertible Preferred Stock so as to affect them adversely. |
Fractional Shares. No fractional shares of common stock will be issued upon conversion of shares of our Series A Preferred Stock. Rather, we shall round up to the next whole share.
Royalties. Pursuant to the terms of the Certificate of Designations for our outstanding Series A Preferred Stock, we may be required to pay, subject to certain vesting requirements, in the aggregate, a royalty equal to (i) 4.5% of Net Sales (as defined in the Certificate of Designation) from MAT 2203 and/or MAT 2501, subject in all cases to a cap of $25 million per calendar year, and (ii) 7.5% of Licensing Proceeds (as defined in the Certificate of Designations) from MAT2203 and/or MAT2501, subject in all cases to a cap of $10 million per calendar year. Our obligation to pay such royalty will expire when the patents covering the applicable product expire, which is currently expected to be in 2033.
Royalty Vesting. The shares of Series A Preferred Stock will vest, in equal thirds, upon each of the first, second and third vesting dates; provided however, if the Series A Preferred Stock automatically convert into common stock prior to July 29, 2019, which is the 36 month anniversary of the initial closing of the 2016 Private Placement, then the outstanding shares of Series A Preferred Stock shall be deemed to be fully vested as of the date of conversion. Once a vesting date has occurred, the portion that vested on such vesting date shall be unaffected by any conversion thereafter of such shares of our Series A Preferred Stock.
Each holder is entitled to a portion of the royalty payment, if any, as determined based on the length of time that such holder’s shares of Series A Preferred Stock remain unconverted. If a holder elects to convert all of his shares of Series A Preferred Stock into common stock prior to July 29, 2017, the first 12 month anniversary after the Initial Closing, the holder will forfeit any rights to future royalty payments, if any. If a holder elects to convert any portion of his Series A Preferred Stock to common stock at any time prior to the third vesting date, such holder will forfeit any rights to future royalty payments if any, with respect to such the unvested portion of such converted shares.
Allocation of Royalty Payment. Once the aggregate Royalty Payment Amount is calculated based on the criteria set forth above under “Royalties,” that amount will be allocated to the holders of the Participating Royalty Interests (as defined in the Certificate of Designations) based on their pro rata ownership. The royalty payable to each holder shall be calculated as follows:
(i) Prior to the third Vesting Date, the royalty payable to each holder will be equal to the aggregate Royalty Payment Amount divided by the aggregate Participating Royalty Interests on the applicable record date multiplied by the number of Participating Royalty Interests held by such holder on the applicable record date.
(ii) On or after the third Vesting Date, the Royalty payable to each holder will be calculated by multiplying the aggregate Royalty Payment Amount by the percentage set forth in each holder’s Royalty Payment Rights certificate. The percentage set forth in each Royalty Payment Rights certificate will be calculated as follows:
Number of Participating Royalty Interests Held by Investor on the Third Vesting Date
Total Participating Royalty Interests on the Third Vesting Date
Separability. The royalty payment rights may not be transferred separately from the Series A Preferred Stock until July 29, 2019. Prior to July 29, 2019, if a holder transfers any of its shares of Series A Preferred Stock, such holder will lose any rights to any future royalty payments with respect to the shares of Series A Preferred Stock that were transferred. After July 29, 2019, we will issue a certificate representing the royalty payment rights to (i) each holder of Series A Preferred Stock at such 36 month anniversary date and (ii) any holder of Series A Preferred Stock which converted its shares of Series A Preferred Stock prior to July 29, 2019 but is listed on a list of holders as at a vesting date as a person entitled to receive royalty payments. Following the issuance of royalty payment rights certificates, such royalty payment rights may be transferred, subject to the availability of an exemption from registration under applicable state and federal securities laws, separately from the Series A Preferred Stock.
| 8 |
Unsecured Obligations. The royalty payment rights are unsecured obligations of ours.
VStock Transfer, LLC is the transfer agent and registrar for our Series A Preferred Stock.
Anti-takeover Effects of Delaware Law and of our Amended and Restated Certificate of Incorporation
The following paragraphs summarize certain provisions of the DGCL and our amended and restated certificate of incorporation that may have the effect of discouraging an acquisition of Matinas. The summary does not purport to be complete and is subject to and qualified in its entirety by reference to the DGCL and our amended and restated certificate of incorporation and by-laws, copies of which are on file with the SEC. Please refer to “Additional Information” below for directions on obtaining these documents.
Section 203 of the Delaware General Corporation Law
We are subject to Section 203 of the Delaware General Corporation Law, which prohibits a Delaware corporation from engaging in any business combination with any interested stockholder for a period of three years after the date that such stockholder became an interested stockholder, with the following exceptions:
| ● | before such date, the board of directors of the corporation approved either the business combination or the transaction that resulted in the stockholder becoming an interested stockholder; |
| ● | upon completion of the transaction that resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction began, excluding for purposes of determining the voting stock outstanding (but not the outstanding voting stock owned by the interested stockholder) those shares owned (i) by persons who are directors and also officers and (ii) employee stock plans in which employee participants do not have the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange offer; or |
| ● | on or after such date, the business combination is approved by the board of directors and authorized at an annual or special meeting of the stockholders, and not by written consent, by the affirmative vote of at least 66 2/3% of the outstanding voting stock that is not owned by the interested stockholder. |
In general, Section 203 defines business combination to include the following:
| ● | any merger or consolidation involving the corporation and the interested stockholder; |
| ● | any sale, transfer, pledge or other disposition of 10% or more of the assets of the corporation involving the interested stockholder; |
| ● | subject to certain exceptions, any transaction that results in the issuance or transfer by the corporation of any stock of the corporation to the interested stockholder; |
| ● | any transaction involving the corporation that has the effect of increasing the proportionate share of the stock or any class or series of the corporation beneficially owned by the interested stockholder; or |
| ● | the receipt by the interested stockholder of the benefit of any loss, advances, guarantees, pledges or other financial benefits by or through the corporation. |
In general, Section 203 defines an “interested stockholder” as an entity or person who, together with the person’s affiliates and associates, beneficially owns, or within three years prior to the time of determination of interested stockholder status did own, 15% or more of the outstanding voting stock of the corporation.
| 9 |
Certificate of Incorporation and Bylaws
Our certificate of incorporation and bylaws contain provisions that could have the effect of discouraging potential acquisition proposals or tender offers or delaying or preventing a change of control of our company. These provisions are as follows:
| ● | they provide that special meetings of stockholders may be called only by the board of directors, President or our Chairman of the Board of Directors, or at the request in writing by stockholders of record owning at least fifty (50%) percent of the issued and outstanding voting shares of common stock; |
| ● | they do not include a provision for cumulative voting in the election of directors. Under cumulative voting, a minority stockholder holding a sufficient number of shares may be able to ensure the election of one or more directors. The absence of cumulative voting may have the effect of limiting the ability of minority stockholders to effect changes in our board of directors; and |
| ● | they allow us to issue “blank check” preferred stock, the terms of which may be established and shares of which may be issued without stockholder approval. |
Potential Effects of Authorized but Unissued Stock
We have shares of common stock and preferred stock available for future issuance without stockholder approval. We may utilize these additional shares for a variety of corporate purposes, including future public offerings to raise additional capital, to facilitate corporate acquisitions or payment as a dividend on the capital stock.
The existence of unissued and unreserved common stock and preferred stock may enable our board of directors to issue shares to persons friendly to current management or to issue preferred stock with terms that could render more difficult or discourage a third-party attempt to obtain control of us by means of a merger, tender offer, proxy contest or otherwise, thereby protecting the continuity of our management. In addition, the board of directors has the discretion to determine designations, rights, preferences, privileges and restrictions, including voting rights, dividend rights, conversion rights, redemption privileges and liquidation preferences of each series of preferred stock, all to the fullest extent permissible under the DGCL and subject to any limitations set forth in our amended and restated certificate of incorporation. The purpose of authorizing the board of directors to issue preferred stock and to determine the rights and preferences applicable to such preferred stock is to eliminate delays associated with a stockholder vote on specific issuances. The issuance of preferred stock, while providing desirable flexibility in connection with possible financings, acquisitions and other corporate purposes, could have the effect of making it more difficult for a third-party to acquire, or could discourage a third-party from acquiring, a majority of our outstanding voting stock.
We summarize below some of the provisions that will apply to the warrants unless the applicable prospectus supplement provides otherwise. This summary may not contain all information that is important to you. The complete terms of the warrants will be contained in the applicable warrant certificate and warrant agreement. These documents have been or will be included or incorporated by reference as exhibits to the registration statement of which this prospectus is a part. You should read the warrant certificate and the warrant agreement. You should also read the prospectus supplement, which will contain additional information and which may update or change some of the information below.
General
We may issue, together with common or preferred stock as units or separately, warrants for the purchase of shares of our common stock, shares of our preferred stock, debt securities or other securities. The terms of each warrant will be discussed in the applicable prospectus supplement relating to the particular series of warrants. The form(s) of certificate representing the warrants and/or the warrant agreement, will be, in each case, filed with the SEC as an exhibit to a document incorporated by reference in the registration statement of which this prospectus is a part on or prior to the date of any prospectus supplement relating to an offering of the particular warrant. The following summary of material provisions of the warrants and the warrant agreements are subject to, and qualified in their entirety by reference to, all the provisions of the warrant agreement and warrant certificate applicable to a particular series of warrants.
| 10 |
The prospectus supplement relating to any series of warrants that are offered by this prospectus will describe, among other things, the following terms to the extent they are applicable to that series of warrants:
| ● | the procedures and conditions relating to the exercise of the warrants; |
| ● | the number of shares of our common or preferred stock, if any, issued with the warrants; |
| ● | the date, if any, on and after which the warrants and any related shares of our common or preferred stock will be separately transferable; |
| ● | the offering price of the warrants, if any; |
| ● | the number of shares of our common or preferred stock, debt securities or other securities which may be purchased upon exercise of the warrants and the price or prices at which such securities may be purchased upon exercise; |
| ● | the date on which the right to exercise the warrants will begin and the date on which the right will expire; |
| ● | a discussion of any material United States federal income tax considerations applicable to the exercise of the warrants; |
| ● | anti-dilution provisions of the warrants, if any; |
| ● | call provisions of the warrants, if any; and |
| ● | any other material terms of the warrants. |
Each warrant may entitle the holder to purchase for cash, or, in limited circumstances, by effecting a cashless exercise for, the number of shares of our common or preferred stock at the exercise price that is described in the applicable prospectus supplement. Warrants will be exercisable during the period of time described in the applicable prospectus supplement. After that period, unexercised warrants will be void. Warrants may be exercised in the manner described in the applicable prospectus supplement.
A holder of a warrant will not have any of the rights of a holder of our common or preferred stock before the stock is purchased upon exercise of the warrant. Therefore, before a warrant is exercised, the holder of the warrant will not be entitled to receive any dividend payments or exercise any voting or other rights associated with shares of our common or preferred stock which may be purchased when the warrant is exercised.
Transfer Agent and Registrar
The transfer agent and registrar, if any, for any warrants will be set forth in the applicable prospectus supplement.
| 11 |
DESCRIPTION OF DEBT SECURITIES
We summarize below some of the provisions that will apply to the debt securities unless the applicable prospectus supplement provides otherwise. This summary may not contain all information that is important to you. The debt securities may be issued pursuant to, in the case of senior debt securities, a senior indenture, and in the case of subordinated debt securities, a subordinated indenture, in each case in the forms filed as exhibits to this registration statement, which we refer to as the “indentures.” The indentures will be entered into between us and a trustee to be named prior to the issuance of any debt securities, which we refer to as the “trustee.” The indentures will not limit the amount of debt securities that can be issued thereunder and will provide that the debt securities may be issued from time to time in one or more series pursuant to the terms of one or more securities resolutions or supplemental indentures creating such series.
The complete terms of the debt securities will be contained in the applicable indenture for the particular offering of debt securities itself which will describe the terms and definitions of the offered debt securities and contain additional information about such debt securities. You should also read the prospectus supplement, which will contain additional information and which may update or change some of the information below.
General
When we offer to sell a particular series of debt securities, we will describe the specific terms of the securities in a prospectus supplement. The prospectus supplement will set forth the following terms, as applicable, of the debt securities offered thereby:
| ● | the designation, aggregate principal amount, currency or composite currency and denominations; |
| ● | the price at which such debt securities will be issued and, if an index formula or other method is used, the method for determining amounts of principal or interest; |
| ● | the maturity date and other dates, if any, on which principal will be payable; |
| ● | whether or not the debt securities will be secured or unsecured, and the terms of any secured debt; |
| ● | whether the debt securities rank as senior debt, senior subordinated debt, subordinated debt or any combination thereof, and the terms of any subordination; |
| ● | the interest rate (which may be fixed or variable), if any; |
| ● | the date or dates from which interest will accrue and on which interest will be payable, and the record dates for the payment of interest; |
| ● | the manner of paying principal and interest; |
| ● | the place or places where principal and interest will be payable; |
| ● | the terms of any mandatory or optional redemption by us or any third party including any sinking fund; |
| ● | the terms of any conversion or exchange; |
| ● | the terms of any redemption at the option of holders or put by the holders; |
| ● | any tax indemnity provisions; |
| ● | if the debt securities provide that payments of principal or interest may be made in a currency other than that in which debt securities are denominated, the manner for determining such payments; |
| ● | the portion of principal payable upon acceleration of a Discounted Debt Security (as defined below); |
| ● | whether and upon what terms debt securities may be defeased; |
| 12 |
| ● | any events of default or covenants in addition to or in lieu of those set forth in the indentures; |
| ● | provisions for electronic issuance of debt securities or for debt securities in uncertificated form; and |
| ● | any additional provisions or other special terms not inconsistent with the provisions of the indentures, including any terms that may be required or advisable under United States or other applicable laws or regulations, or advisable in connection with the marketing of the debt securities. |
Debt securities of any series may be issued as registered debt securities or uncertificated debt securities, in such denominations as specified in the terms of the series.
Securities may be issued under the indentures as Discounted Debt Securities to be offered and sold at a substantial discount from the principal amount thereof. Special United States federal income tax and other considerations applicable thereto will be described in the prospectus supplement relating to such Discounted Debt Securities. “Discounted Debt Security” means a security where the amount of principal due upon acceleration is less than the stated principal amount.
We are not obligated to issue all debt securities of one series at the same time and, unless otherwise provided in the prospectus supplement, we may reopen a series, without the consent of the holders of the debt securities of that series, for the issuance of additional debt securities of that series. Additional debt securities of a particular series will have the same terms and conditions as outstanding debt securities of such series, except for the date of original issuance and the offering price, and will be consolidated with, and form a single series with, such outstanding debt securities.
Ranking
The senior debt securities will rank equally with all of our other senior and unsubordinated debt. Our secured debt, if any, will be effectively senior to the senior debt securities to the extent of the value of the assets securing such debt. The subordinated debt securities will be subordinate and junior in right of payment to all of our present and future senior indebtedness to the extent and in the manner described in the prospectus supplement and as set forth in the board resolution, officer’s certificate or supplemental indenture relating to such offering.
We have only a stockholder’s claim on the assets of our subsidiaries. This stockholder’s claim is junior to the claims that creditors of our subsidiaries have against our subsidiaries. Holders of our debt securities will be our creditors and not creditors of any of our subsidiaries. As a result, all the existing and future liabilities of our subsidiaries, including any claims of their creditors, will effectively be senior to the debt securities with respect to the assets of our subsidiaries. In addition, to the extent that we issue any secured debt, the debt securities will be effectively subordinated to such secured debt to the extent of the value of the assets securing such secured debt.
The debt securities will be obligations exclusively of Matinas BioPharma Holdings, Inc. To the extent that our ability to service our debt, including the debt securities, may be dependent upon the earnings of our subsidiaries, our ability to do so will be dependent on the ability of our subsidiaries to distribute those earnings to us as dividends, loans or other payments.
Certain Covenants
Any covenants that may apply to a particular series of debt securities will be described in the prospectus supplement relating thereto.
Successor Obligor
The indentures provide that, unless otherwise specified in the securities resolution or supplemental indenture establishing a series of debt securities, we shall not consolidate with or merge into, or transfer all or substantially all of our assets to, any person in any transaction in which we are not the survivor, unless:
| ● | the person is organized under the laws of the United States or a jurisdiction within the United States; |
| ● | the person assumes by supplemental indenture all of our obligations under the relevant indenture, the debt securities and any coupons; |
| 13 |
| ● | immediately after the transaction no Default (as defined below) exists; and |
| ● | we deliver to the trustee an officers’ certificate and opinion of counsel stating that the transaction complies with the foregoing requirements. |
In such event, the successor will be substituted for us, and thereafter all of our obligations under the relevant indenture, the debt securities and any coupons will terminate.
Exchange of Debt Securities
Registered debt securities may be exchanged for an equal aggregate principal amount of registered debt securities of the same series and date of maturity in such authorized denominations as may be requested upon surrender of the registered debt securities at an agency of the Company maintained for such purpose and upon fulfillment of all other requirements of such agent.
Defaults and Remedies
Unless the securities resolution or supplemental indenture establishing the series otherwise provides (in which event the prospectus supplement will so state), an “Event of Default” with respect to a series of debt securities will occur if:
| (1) | we default in any payment of interest on any debt securities of such series when the same becomes due and payable and the default continues for a period of 30 days; |
| (2) | we default in the payment of the principal and premium, if any, of any debt securities of such series when the same becomes due and payable at maturity or upon redemption, acceleration or otherwise and such default shall continue for five or more days; |
| (3) | we default in the performance of any of our other agreements applicable to the series and the default continues for 30 days after the notice specified below; |
| (4) | a court of competent jurisdiction enters an order or decree under any Bankruptcy Law (as defined below) that: |
| (A) | is for relief against us in an involuntary case, |
| (B) | appoints a Custodian (as defined below) for us or for all or substantially all of our property, or |
| (C) | orders the liquidation of us, and the order or decree remains unstayed and in effect for 90 days; |
| (5) | we pursuant to or within the meaning of any Bankruptcy Law: |
| (A) | commence a voluntary case, |
| (B) | consent to the entry of an order for relief against us in an involuntary case, |
| (C) | consent to the appointment of a Custodian for us or for all or substantially all of our property, or |
| (D) | make a general assignment for the benefit of our creditors; or |
| (6) | there occurs any other Event of Default provided for in such series. |
The term “Bankruptcy Law” means Title 11 of the United States Code or any similar Federal or State law for the relief of debtors. The term “Custodian” means any receiver, trustee, assignee, liquidator or a similar official under any Bankruptcy Law.
“Default” means any event which is, or after notice or passage of time would be, an Event of Default. A Default under subparagraph (3) above is not an Event of Default until the trustee or the holders of at least 25% in principal amount of the series notify us of the Default and we do not cure the Default within the time specified after receipt of the notice.
| 14 |
The trustee may require indemnity satisfactory to it before it enforces the indentures or the debt securities of the series. Subject to certain limitations, holders of a majority in principal amount of the debt securities of the series may direct the trustee in its exercise of any trust or power with respect to such series. Except in the case of Default in payment on a series, the trustee may withhold from securityholders of such series notice of any continuing Default if the trustee determines that withholding notice is in the interest of such securityholders. We are required to furnish the trustee annually a brief certificate as to our compliance with all conditions and covenants under the indentures.
The indentures do not have cross-default provisions. Thus, a default by us on any other debt, including any other series of debt securities, would not constitute an Event of Default.
Amendments and Waivers
The indentures and the debt securities or any coupons of the series may be amended, and any Default may be waived as follows:
Unless the securities resolution or supplemental indenture otherwise provides (in which event the applicable prospectus supplement will so state), the debt securities and the indentures may be amended with the consent of the holders of a majority in principal amount of the debt securities of all series affected voting as one class. Unless the securities resolution or supplemental indenture otherwise provides (in which event the applicable prospectus supplement will so state), a Default other than a Default in payment on a particular series may be waived with the consent of the holders of a majority in principal amount of the debt securities of the series. However, without the consent of each securityholder affected, no amendment or waiver may:
| ● | change the fixed maturity of or the time for payment of interest on any debt security; |
| ● | reduce the principal, premium or interest payable with respect to any debt security; |
| ● | change the place of payment of a debt security or the currency in which the principal or interest on a debt security is payable; |
| ● | change the provisions for calculating any redemption or repurchase price with respect to any debt security; |
| ● | reduce the amount of debt securities whose holders must consent to an amendment or waiver; |
| ● | make any change that materially adversely affects the right to convert any debt security; |
| ● | waive any Default in payment of principal of or interest on a debt security; or |
| ● | adversely affect any holder’s rights with respect to redemption or repurchase of a debt security. |
Without the consent of any securityholder, the indentures or the debt securities may be amended to:
| ● | provide for assumption of our obligations to securityholders in the event of a merger or consolidation requiring such assumption; |
| ● | to cure any ambiguity, omission, defect or inconsistency; |
| ● | to conform the terms of the debt securities to the description thereof in the prospectus and prospectus supplement offering such debt securities; |
| ● | to create a series and establish its terms; |
| ● | to provide for assumption of our obligations to securityholders in the event of a merger or consolidation requiring such assumption; |
| ● | to make any change that does not adversely affect the rights of any securityholder; |
| ● | to add to our covenants; or |
| ● | to make any other change to the indentures so long as no debt securities are outstanding. |
| 15 |
Conversion Rights
Any securities resolution or supplemental indenture establishing a series of debt securities may provide that the debt securities of such series will be convertible at the option of the holders thereof into or for our common stock or other equity or debt instruments. The securities resolution or supplemental indenture may establish, among other things, (1) the number or amount of shares of common stock or other equity or debt instruments for which $1,000 aggregate principal amount of the debt securities of the series is convertible, as may be adjusted pursuant to the terms of the relevant indenture and the securities resolution; and (2) provisions for adjustments to the conversion rate and limitations upon exercise of the conversion right. The indentures provide that we will not be required to make an adjustment in the conversion rate unless the adjustment would require a cumulative change of at least 1% in the conversion rate. However, we will carry forward any adjustments that are less than 1% of the conversion rate and take them into account in any subsequent adjustment of the conversion rate.
Legal Defeasance and Covenant Defeasance
Debt securities of a series may be defeased in accordance with their terms and, unless the securities resolution or supplemental indenture establishing the terms of the series otherwise provides, as set forth below. We at any time may terminate as to a series all of our obligations (except for certain obligations, including obligations with respect to the defeasance trust and obligations to register the transfer or exchange of a debt security, to replace destroyed, lost or stolen debt securities and coupons and to maintain paying agencies in respect of the debt securities) with respect to the debt securities of the series and any related coupons and the relevant indenture, which we refer to as legal defeasance. We at any time may terminate as to a series our obligations with respect to any restrictive covenants which may be applicable to a particular series, which we refer to as covenant defeasance.
We may exercise our legal defeasance option notwithstanding our prior exercise of our covenant defeasance option. If we exercise our legal defeasance option, a series may not be accelerated because of an Event of Default. If we exercise our covenant defeasance option, a series may not be accelerated by reference to any covenant which may be applicable to a series.
To exercise either defeasance option as to a series, we must (1) irrevocably deposit in trust with the trustee (or another trustee) money or U.S. Government Obligations (as defined below), deliver a certificate from a nationally recognized firm of independent accountants expressing their opinion that the payments of principal and interest when due on the deposited U.S. Government Obligations, without reinvestment, plus any deposited money without investment will provide cash at such times and in such amounts as will be sufficient to pay the principal and interest when due on all debt securities of such series to maturity or redemption, as the case may be; and (2) comply with certain other conditions. In particular, we must obtain an opinion of tax counsel that the defeasance will not result in recognition of any gain or loss to holders for federal income tax purposes.
“U.S. Government Obligations” means direct obligations of the United States or any agency or instrumentality of the United States, the payment of which is unconditionally guaranteed by the United States, which, in either case, have the full faith and credit of the United States pledged for payment and which are not callable at the issuer’s option, or certificates representing an ownership interest in such obligations.
| 16 |
Regarding the Trustee
Unless otherwise indicated in a prospectus supplement, the trustee will also act as depository of funds, transfer agent, paying agent and conversion agent, as applicable, with respect to the debt securities. We may remove the trustee as the trustee under a given indenture with or without cause if we so notify the trustee three months in advance and if no Default occurs during the three-month period. The indenture trustee may also provide additional unrelated services to us as a depository of funds, registrar, trustee and similar services.
Governing Law
The indentures and the debt securities will be governed by New York law, except to the extent that the Trust Indenture Act of 1939 is applicable.
DESCRIPTION OF SUBSCRIPTION RIGHTS
We may issue subscription rights to purchase our common stock, preferred stock or debt securities. These subscription rights may be offered independently or together with any other security offered hereby and may or may not be transferable by the stockholder receiving the subscription rights in such offering. In connection with any offering of subscription rights, we may enter into a standby arrangement with one or more underwriters or other purchasers pursuant to which the underwriters or other purchasers may be required to purchase any securities remaining unsubscribed for after such offering.
The prospectus supplement relating to any subscription rights we offer, if any, will, to the extent applicable, include specific terms relating to the offering, including some or all of the following:
| ● | the price, if any, for the subscription rights; |
| ● | the exercise price payable for our common stock, preferred stock or debt securities upon the exercise of the subscription rights; |
| ● | the number of subscription rights to be issued to each stockholder; |
| ● | the number and terms of our common stock, preferred stock or debt securities which may be purchased per each subscription right; |
| ● | the extent to which the subscription rights are transferable; |
| ● | any other terms of the subscription rights, including the terms, procedures and limitations relating to the exchange and exercise of the subscription rights; |
| ● | the date on which the right to exercise the subscription rights shall commence, and the date on which the subscription rights shall expire; |
| ● | the extent to which the subscription rights may include an over-subscription privilege with respect to unsubscribed securities or an over-allotment privilege to the extent the securities are fully subscribed; and |
| ● | if applicable, the material terms of any standby underwriting or purchase arrangement which may be entered into by Matinas in connection with the offering of subscription rights. |
We may issue units comprised of one or more of the other securities described in this prospectus in any combination. Each unit will be issued so that the holder of the unit is also the holder of each security included in the unit. Thus, the holder of a unit will have the rights and obligations of a holder of each included security (but, to the extent convertible securities are included in the units, the holder of the units will be deemed the holder of the convertible securities and not the holder of the underlying securities). The unit agreement under which a unit is issued may provide that the securities included in the unit may not be held or transferred separately, at any time or at any time before a specified date. The applicable prospectus supplement may describe:
| 17 |
| ● | the designation and terms of the units and of the securities comprising the units, including whether and under what circumstances those securities may be held or transferred separately; |
| ● | any provisions for the issuance, payment, settlement, transfer or exchange of the units or of the securities comprising the units; |
| ● | the terms of the unit agreement governing the units; |
| ● | United States federal income tax considerations relevant to the units; and |
| ● | whether the units will be issued in fully registered global form. |
This summary of certain general terms of units and any summary description of units in the applicable prospectus supplement do not purport to be complete and are qualified in their entirety by reference to all provisions of the applicable unit agreement and, if applicable, collateral arrangements and depositary arrangements relating to such units. The forms of the unit agreements and other documents relating to a particular issue of units will be filed with the SEC each time we issue units, and you should read those documents for provisions that may be important to you.
FORMS OF SECURITIES
Each debt security and, to the extent applicable, warrant, subscription right and unit, will be represented either by a certificate issued in definitive form to a particular investor or by one or more global securities representing the entire issuance of securities. Certificated securities in definitive form and global securities will be issued in registered form. Definitive securities name you or your nominee as the owner of the security, and in order to transfer or exchange these securities or to receive payments other than interest or other interim payments, you or your nominee must physically deliver the securities to the trustee, registrar, paying agent or other agent, as applicable. Global securities name a depositary or its nominee as the owner of the debt securities or warrants represented by these global securities. The depositary maintains a computerized system that will reflect each investor’s beneficial ownership of the securities through an account maintained by the investor with its broker/dealer, bank, trust company or other representative, as we explain more fully below.
Global Securities
Registered Global Securities. We may issue the registered debt securities and, to the extent applicable, warrants, subscription rights and units in the form of one or more fully registered global securities that will be deposited with a depositary or its nominee identified in the applicable prospectus supplement and registered in the name of that depositary or nominee. In those cases, one or more registered global securities will be issued in a denomination or aggregate denominations equal to the portion of the aggregate principal or face amount of the securities to be represented by registered global securities. Unless and until it is exchanged in whole for securities in definitive registered form, a registered global security may not be transferred except as a whole by and among the depositary for the registered global security, the nominees of the depositary or any successors of the depositary or those nominees.
If not described below, any specific terms of the depositary arrangement with respect to any securities to be represented by a registered global security will be described in the prospectus supplement relating to those securities. We anticipate that the following provisions will apply to all depositary arrangements.
Ownership of beneficial interests in a registered global security will be limited to persons, called participants, that have accounts with the depositary or persons that may hold interests through participants. Upon the issuance of a registered global security, the depositary will credit, on its book-entry registration and transfer system, the participants’ accounts with the respective principal or face amounts of the securities beneficially owned by the participants. Any dealers, underwriters or agents participating in the distribution of the securities will designate the accounts to be credited. Ownership of beneficial interests in a registered global security will be shown on, and the transfer of ownership interests will be effected only through, records maintained by the depositary, with respect to interests of participants, and on the records of participants, with respect to interests of persons holding through participants. The laws of some states may require that some purchasers of securities take physical delivery of these securities in definitive form. These laws may impair your ability to own, transfer or pledge beneficial interests in registered global securities.
| 18 |
So long as the depositary, or its nominee, is the registered owner of a registered global security, that depositary or its nominee, as the case may be, will be considered the sole owner or holder of the securities represented by the registered global security for all purposes under the applicable indenture or warrant agreement. Except as described below, owners of beneficial interests in a registered global security will not be entitled to have the securities represented by the registered global security registered in their names, will not receive or be entitled to receive physical delivery of the securities in definitive form and will not be considered the owners or holders of the securities under the applicable indenture or warrant agreement. Accordingly, each person owning a beneficial interest in a registered global security must rely on the procedures of the depositary for that registered global security and, if that person is not a participant, on the procedures of the participant through which the person owns its interest, to exercise any rights of a holder under the applicable indenture or warrant agreement. We understand that under existing industry practices, if we request any action of holders or if an owner of a beneficial interest in a registered global security desires to give or take any action that a holder is entitled to give or take under the applicable indenture or warrant agreement, the depositary for the registered global security would authorize the participants holding the relevant beneficial interests to give or take that action, and the participants would authorize beneficial owners owning through them to give or take that action or would otherwise act upon the instructions of beneficial owners holding through them.
Principal, premium, if any, and interest payments on debt securities and any payments to holders with respect to warrants represented by a registered global security registered in the name of a depositary or its nominee will be made to the depositary or its nominee, as the case may be, as the registered owner of the registered global security. None of Matinas, the trustees, the warrant agents or any other agent of Matinas, agent of the trustees or agent of the warrant agents will have any responsibility or liability for any aspect of the records relating to payments made on account of beneficial ownership interests in the registered global security or for maintaining, supervising or reviewing any records relating to those beneficial ownership interests.
We expect that the depositary for any of the securities represented by a registered global security, upon receipt of any payment of principal, premium, interest or other distribution of underlying securities or other property to holders on that registered global security, will immediately credit participants’ accounts in amounts proportionate to their respective beneficial interests in that registered global security as shown on the records of the depositary. We also expect that payments by participants to owners of beneficial interests in a registered global security held through participants will be governed by standing customer instructions and customary practices, as is now the case with the securities held for the accounts of customers in bearer form or registered in “street name,” and will be the responsibility of those participants.
If the depositary for any of these securities represented by a registered global security is at any time unwilling or unable to continue as depositary or ceases to be a clearing agency registered under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and a successor depositary registered as a clearing agency under the Exchange Act is not appointed by us within 90 days, we will issue securities in definitive form in exchange for the registered global security that had been held by the depositary. Any securities issued in definitive form in exchange for a registered global security will be registered in the name or names that the depositary gives to the relevant trustee or warrant agent or other relevant agent of ours or theirs. It is expected that the depositary’s instructions will be based upon directions received by the depositary from participants with respect to ownership of beneficial interests in the registered global security that had been held by the depositary.
Initial Offering and Sale of Securities
Unless otherwise set forth in a prospectus supplement accompanying this prospectus, we, and certain holders of our securities, may sell the securities being offered hereby, from time to time, by one or more of the following methods:
| ● | to or through underwriting syndicates represented by managing underwriters; |
| ● | through one or more underwriters without a syndicate for them to offer and sell to the public; |
| ● | through dealers or agents; and |
| ● | to investors directly in negotiated sales or in competitively bid transactions. |
Offerings of securities covered by this prospectus also may be made into an existing trading market for those securities in transactions at other than a fixed price, either:
| ● | on or through the facilities of the NYSE MKT or any other securities exchange or quotation or trading service on which those securities may be listed, quoted, or traded at the time of sale; and/or |
| 19 |
| ● | to or through a market maker otherwise than on the securities exchanges or quotation or trading services set forth above. |
At-the-market offerings, if any, will be conducted by underwriters acting as principal or agent of the Company, who may also be third-party sellers of securities as described above. The prospectus supplement with respect to the offered securities will set forth the terms of the offering of the offered securities, including:
| ● | the name or names of any underwriters, dealers or agents; |
| ● | the purchase price of the offered securities and the proceeds to us from such sale; |
| ● | any underwriting discounts and commissions or agency fees and other items constituting underwriters’ or agents’ compensation; |
| ● | any initial public offering price and any discounts or concessions allowed or reallowed or paid to dealers; |
| ● | any securities exchange on which such offered securities may be listed; and |
| ● | any underwriter, agent or dealer involved in the offer and sale of any series of the securities will be named in the prospectus supplement. |
The distribution of the securities may be effected from time to time in one or more transactions:
| ● | at fixed prices, which may be changed; |
| ● | at market prices prevailing at the time of the sale; |
| ● | at varying prices determined at the time of sale; or |
| ● | at negotiated prices. |
Each prospectus supplement will set forth the manner and terms of an offering of securities including:
| ● | whether that offering is being made to underwriters or through agents or directly; |
| ● | the rules and procedures for any auction or bidding process, if used; |
| ● | the securities’ purchase price or initial public offering price; and |
| ● | the proceeds we anticipate from the sale of the securities, if any. |
In addition, we may enter into derivative or hedging transactions with third parties, or sell securities not covered by this prospectus to third parties in privately negotiated transactions. If the applicable prospectus supplement indicates, in connection with such a transaction, the third parties may sell securities covered by and pursuant to this prospectus and an applicable prospectus supplement. If so, the third party may use securities pledged by us or borrowed from us or others to settle such sales and may use securities received from us to close out any related short positions. We may also loan or pledge securities covered by this prospectus and an applicable prospectus supplement to third parties, who may sell the loaned securities or, in an event of default in the case of a pledge, sell the pledged securities pursuant to this prospectus and the applicable prospectus supplement.
Sales Through Underwriters
If underwriters are used in the sale of some or all of the securities covered by this prospectus, the underwriters will acquire the securities for their own account. The underwriters may resell the securities, either directly to the public or to securities dealers, at various times in one or more transactions, including negotiated transactions, at a fixed public offering price or at varying prices determined at the time of sale. The obligations of the underwriters to purchase the securities will be subject to certain conditions. Unless indicated otherwise in a prospectus supplement, the underwriters will be obligated to purchase all the securities of the series offered if any of the securities are purchased.
Any initial public offering price and any concessions allowed or reallowed to dealers may be changed intermittently.
| 20 |
Sales Through Agents
Unless otherwise indicated in the applicable prospectus supplement, when securities are sold through an agent, the designated agent will agree, for the period of its appointment as agent, to use its best efforts to sell the securities for our account and will receive commissions from us as will be set forth in the applicable prospectus supplement.
Securities bought in accordance with a redemption or repayment under their terms also may be offered and sold, if so indicated in the applicable prospectus supplement, in connection with a remarketing by one or more firms acting as principals for their own accounts or as agents for us. Any remarketing firm will be identified and the terms of its agreement, if any, with us and its compensation will be described in the prospectus supplement. Remarketing firms may be deemed to be underwriters in connection with the securities remarketed by them.
If so indicated in the applicable prospectus supplement, we may authorize agents, underwriters or dealers to solicit offers by certain specified institutions to purchase securities at a price set forth in the prospectus supplement pursuant to delayed delivery contracts providing for payment and delivery on a future date specified in the prospectus supplement. These contracts will be subject only to those conditions set forth in the applicable prospectus supplement, and the prospectus supplement will set forth the commissions payable for solicitation of these contracts.
Direct Sales
We may also sell offered securities directly to institutional investors or others. In this case, no underwriters or agents would be involved. The terms of such sales will be described in the applicable prospectus supplement.
General Information
Broker-dealers, agents or underwriters may receive compensation in the form of discounts, concessions or commissions from us and/or the purchasers of securities for whom such broker-dealers, agents or underwriters may act as agents or to whom they sell as principal, or both (this compensation to a particular broker-dealer might be in excess of customary commissions).
Underwriters, dealers and agents that participate in any distribution of the offered securities may be deemed “underwriters” within the meaning of the Securities Act, so any discounts or commissions they receive in connection with the distribution may be deemed to be underwriting compensation. Those underwriters and agents may be entitled, under their agreements with us, to indemnification by us against certain civil liabilities, including liabilities under the Securities Act, or to contribution by us to payments that they may be required to make in respect of those civil liabilities. Certain of those underwriters or agents may be customers of, engage in transactions with, or perform services for, us or our affiliates in the ordinary course of business. We will identify any underwriters or agents, and describe their compensation, in a prospectus supplement. Any institutional investors or others that purchase offered securities directly, and then resell the securities, may be deemed to be underwriters, and any discounts or commissions received by them from us and any profit on the resale of the securities by them may be deemed to be underwriting discounts and commissions under the Securities Act.
We will file a supplement to this prospectus, if required, pursuant to Rule 424(b) under the Securities Act, if we enter into any material arrangement with a broker, dealer, agent or underwriter for the sale of securities through a block trade, special offering, exchange distribution or secondary distribution or a purchase by a broker or dealer. Such prospectus supplement will disclose:
| ● | the name of any participating broker, dealer, agent or underwriter; |
| ● | the number and type of securities involved; |
| ● | the price at which such securities were sold; |
| ● | any securities exchanges on which such securities may be listed; |
| ● | the commissions paid or discounts or concessions allowed to any such broker, dealer, agent or underwriter where applicable; and |
| ● | other facts material to the transaction. |
| 21 |
In order to facilitate the offering of certain securities under this prospectus or an applicable prospectus supplement, certain persons participating in the offering of those securities may engage in transactions that stabilize, maintain or otherwise affect the price of those securities during and after the offering of those securities. Specifically, if the applicable prospectus supplement permits, the underwriters of those securities may over-allot or otherwise create a short position in those securities for their own account by selling more of those securities than have been sold to them by us and may elect to cover any such short position by purchasing those securities in the open market.
In addition, the underwriters may stabilize or maintain the price of those securities by bidding for or purchasing those securities in the open market and may impose penalty bids, under which selling concessions allowed to syndicate members or other broker-dealers participating in the offering are reclaimed if securities previously distributed in the offering are repurchased in connection with stabilization transactions or otherwise. The effect of these transactions may be to stabilize or maintain the market price of the securities at a level above that which might otherwise prevail in the open market. The imposition of a penalty bid may also affect the price of securities to the extent that it discourages resales of the securities. No representation is made as to the magnitude or effect of any such stabilization or other transactions. Such transactions, if commenced, may be discontinued at any time.
In order to comply with the securities laws of certain states, if applicable, the securities must be sold in such jurisdictions only through registered or licensed brokers or dealers. In addition, in certain states the securities may not be sold unless they have been registered or qualified for sale in the applicable state or an exemption from the registration or qualification requirement is available and is complied with.
Rule 15c6-1 under the Securities Exchange Act of 1934 generally requires that trades in the secondary market settle in three business days, unless the parties to any such trade expressly agree otherwise. Your prospectus supplement may provide that the original issue date for your securities may be more than three scheduled business days after the trade date for your securities. Accordingly, in such a case, if you wish to trade securities on any date prior to the third business day before the original issue date for your securities, you will be required, by virtue of the fact that your securities initially are expected to settle in more than three scheduled business days after the trade date for your securities, to make alternative settlement arrangements to prevent a failed settlement.
This prospectus, any applicable prospectus supplement and any applicable pricing supplement in electronic format may be made available on the Internet sites of, or through other online services maintained by, us and/or one or more of the agents and/or dealers participating in an offering of securities, or by their affiliates. In those cases, prospective investors may be able to view offering terms online and, depending upon the particular agent or dealer, prospective investors may be allowed to place orders online.
Other than this prospectus, any applicable prospectus supplement and any applicable pricing supplement in electronic format, the information on our or any agent’s or dealer’s website and any information contained in any other website maintained by any agent or dealer:
| ● | is not part of this prospectus, any applicable prospectus supplement and any applicable pricing supplement or the registration statement of which they form a part; |
| ● | has not been approved or endorsed by us or by any agent or dealer in its capacity as an agent or dealer, except, in each case, with respect to the respective website maintained by such entity; and |
| ● | should not be relied upon by investors. |
There can be no assurance that we will sell all or any of the securities offered by this prospectus.
This prospectus may also be used in connection with any issuance of any securities upon exercise of a warrant or a subscription right.
| 22 |
In addition, we may issue the securities as a dividend or distribution or in a subscription rights offering to our existing security holders. In some cases, we or dealers acting with us or on our behalf may also purchase securities and reoffer them to the public by one or more of the methods described above. This prospectus may be used in connection with any offering of our securities through any of these methods or other methods described in the applicable prospectus supplement.
Unless otherwise indicated in the applicable prospectus supplement, the validity of the securities offered hereby will be passed upon for us by Lowenstein Sandler LLP, New York, New York. If the validity of the securities offered hereby in connection with offerings made pursuant to this prospectus are passed upon by counsel for the underwriters, dealers or agents, if any, such counsel will be named in the prospectus supplement relating to such offering.
The consolidated balance sheets of Matinas BioPharma Holdings, Inc. and Subsidiaries as of December 31, 2016 and 2015, and the related consolidated statements of operations, stockholders’ equity, and cash flows for each of the years in the two-year period ended December 31, 2016, have been audited by EisnerAmper LLP, independent registered public accounting firm, as stated in their report which is incorporated herein by reference. Such financial statements have been incorporated herein by reference in reliance on the report of such firm given upon their authority as experts in accounting and auditing.
This prospectus is part of a Registration Statement on Form S-3 that we have filed with the SEC relating to the shares of our securities being offered hereby. This prospectus does not contain all of the information in the Registration Statement and its exhibits. The Registration Statement, its exhibits and the documents incorporated by reference in this prospectus and their exhibits, all contain information that is material to the offering of the Securities hereby. Whenever a reference is made in this prospectus to any of our contracts or other documents, the reference may not be complete. You should refer to the exhibits that are a part of the Registration Statement in order to review a copy of the contract or documents. The Registration Statement and the exhibits are available at the SEC’s Public Reference Room or through its Website.
We file annual, quarterly and current reports, proxy statements and other information with the SEC. You can read and copy any materials we file with the SEC at its Public Reference Room at 100 F Street, N.E., Washington, D.C. 20549 and at its regional offices, a list of which is available on the Internet at http://www.sec.gov/contact/addresses.htm. You may obtain information on the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. The SEC maintains an Internet site at http://www.sec.gov that contains reports, proxy and information statements, and other information regarding issuers, such as us, that file electronically with the SEC. Additionally, you may access our filings with the SEC through our website at http://www.Matinasbio.com. The information on our website is not part of this prospectus.
We will provide you without charge, upon your oral or written request, with a copy of any or all reports, proxy statements and other documents we file with the SEC, as well as any or all of the documents incorporated by reference in this prospectus or the registration statement (other than exhibits to such documents unless such exhibits are specifically incorporated by reference into such documents). Requests for such copies should be directed to:
Investor Relations Department
Matinas BioPharma Holdings, Inc.
1545 Route 206 South
Suite 302
Bedminster, NJ 07921
Telephone number: 908-443-1860
| 23 |
You should rely only on the information in this prospectus and the additional information described above and under the heading “Incorporation of Certain Information by Reference” below. We have not authorized any other person to provide you with different information. If anyone provides you with different or inconsistent information, you should not rely upon it. We are not making an offer to sell these securities in any jurisdiction where the offer or sale is not permitted. You should assume that the information in this prospectus was accurate on the date of the front cover of this prospectus only. Our business, financial condition, results of operations and prospects may have changed since that date.
INCORPORATION OF CERTAIN INFORMATION BY REFERENCE
The SEC allows us to “incorporate by reference” information that we file with it into this prospectus, which means that we can disclose important information to you by referring you to those documents. The information incorporated by reference is an important part of this prospectus. The information incorporated by reference is considered to be a part of this prospectus, and information that we file later with the SEC will automatically update and supersede information contained in this prospectus and any accompanying prospectus supplement.
We incorporate by reference the documents listed below that we have previously filed with the SEC:
| ● | our Annual Report on Form 10-K for the fiscal year ended December 31, 2016, filed with the SEC on March 31, 2017; |
| ● | our Current Reports on Form 8-K filed with the SEC on January 19, 2017, and March 30, 2017; and |
| ● | the description of our common stock contained in our Registration Statement on Form 8-A, filed on March 1, 2017, including any amendments thereto or reports filed for the purposes of updating this description. |
All documents filed by us pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act after the date of the initial registration statement and prior to effectiveness of the registration statement shall be deemed to be incorporated by reference in this prospectus and to be a apart hereof from the date of filing of such reports and documents. All reports and other documents that we file with the SEC under Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act after the date of this prospectus but before the termination of the offering of the Securities hereunder will also be considered to be incorporated by reference into this prospectus from the date of the filing of these reports and documents, and will supersede the information herein; provided, however, that all reports that we “furnish” to the SEC will not be considered incorporated by reference into this prospectus. We undertake to provide without charge to each person (including any beneficial owner) who receives a copy of this prospectus, upon written or oral request, a copy of all of the preceding documents that are incorporated by reference (other than exhibits, unless the exhibits are specifically incorporated by reference into these documents). You may request a copy of these materials in the manner set forth under the heading “Additional Information,” above.
| 24 |

27,272,727 Shares of Common Stock
PROSPECTUS SUPPLEMENT
BTIG, LLC
March 15, 2019